Efficacy of a saponin-adjuvanted inactivated respiratory syncytial virus vaccine in calves
- PMID: 15825518
- PMCID: PMC1082864
Efficacy of a saponin-adjuvanted inactivated respiratory syncytial virus vaccine in calves
Abstract
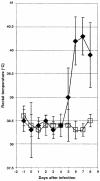
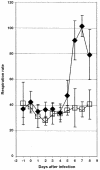
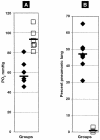
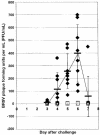
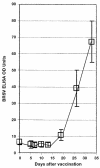
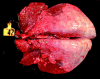
The objective of this study was to determine whether a commercially available, saponin-adjuvanted, inactivated bovine respiratory syncytial virus (BRSV) vaccine would protect calves from experimental infection with virulent BRSV. This was a randomized controlled trial comprising 14, 8- to 9-week-old calves seronegative for BRSV Group 1 calves (n = 8) were not vaccinated and group 2 calves (n = 6) were vaccinated on days 0 and 19 with an inactivated BRSV vaccine. All calves were challenged with virulent BRSV on day 46. Clinical signs, arterial PO2, and immune responses were monitored after challenge. Calves were euthanatized on day 54 (8 d after challenge) and lungs were examined for lesions. Vaccination elicited increases in BRSV-specific immunoglobulin (Ig) G and virus neutralizing antibody titers. Challenge with BRSV resulted in severe respiratory tract disease and extensive pulmonary lesions in control calves, but no signs of clinical disease and minimal or no pulmonary lesions in vaccinated calves. Arterial blood oxygen values on day 53 (7 d after challenge) in control calves were significantly lower than those in vaccinated calves, which remained within normal limits. Control calves shed BRSV for several days after challenge, whereas BRSV was not detected on deep nasal swabs from vaccinated calves. In summary, the results indicated that this inactivated BRSV vaccine provided clinical protection from experimental infection with virulent virus 27 d after vaccination and significantly decreased the prevalence and severity of pulmonary lesions. Efficacy was similar to that reported for other commercial inactivated and modified-live BRSV vaccines.
Résumé — Efficacité d’un vaccin inactif à adjuvant de saponine contre le virus respiratoire syncytial chez le veau. L’objectif de cette étude était de déterminer si un vaccin inactif à adjuvant de saponine contre le virus respiratoire syncytial bovin (VRSB), disponible dans le commerce, pouvait protéger les veaux contre une infection expérimentale par le VRSB virulent. Cette expérience contrôlée comprenait 14 veaux séronégatifs répartis au hasard et âgés de 8 à 9 semaines. Les veaux du groupe 1 (n = 8) n’ont pas été vaccinés et ceux du groupe 2 (n = 6) ont été vaccinés aux jours 0 et 19 avec un vaccin inactivé contre le VRSB. Tous les veaux ont été infectés avec du VRSB virulent au jour 46. Les signes cliniques, le PO2 artériel et les réponses immunitaires ont été enregistrés après l’infection. Les veaux ont été euthanasiés au jour 54 (8 jours après l’infection) et les poumons ont été examinés dans le but d’y observer des lésions. La vaccination a causé une augmentation du titre des immunoglobulines spécifiques au VRSB (Ig)G et des anticorps neutralisants du virus. L’infection par le VRSB a causé une grave maladie du tractus respiratoire et des lésions pulmonaires extensives chez les veaux témoins, mais aucun signe de maladie clinique et pas de lésions ou des lésions minimes seulement chez les veaux vaccinés. Les valeurs de l’oxygène du sang artériel au jour 53 (7 jours après l’infection) chez les veaux témoins étaient significativement plus basses que celles chez les veaux vaccinés, lesquelles se situaient dans les valeurs normales. Les veaux témoins ont éliminé du VRSB pendant plusieurs jours après l’infection alors que les VRSB n’étaient pas détectés dans les prélèvements provenant des voies nasales profondes des veaux vaccinés. En résumé, les résultats indiquent que ce vaccin inactivé contre le VRSB assure une protection clinique contre une infection expérimentale avec le virus virulent 27 jours après la vaccination et diminue significativement la prévalence et la sévérité des lésions pulmonaires. L’efficacité était semblable à celle rapportée pour d’autres vaccins commerciaux inactivés et vivants modifiés contre le VRSB.
(Traduit par Docteur André Blouin)
Figures


Similar articles
-
Efficacy of an inactivated respiratory syncytial virus vaccine in calves.J Am Vet Med Assoc. 2001 Jun 15;218(12):1973-80. doi: 10.2460/javma.2001.218.1973. J Am Vet Med Assoc. 2001. PMID: 11417745 Clinical Trial.
-
Response of calves to challenge exposure with virulent bovine respiratory syncytial virus following intranasal administration of vaccines formulated for parenteral administration.J Am Vet Med Assoc. 2007 Jan 15;230(2):233-43. doi: 10.2460/javma.230.2.233. J Am Vet Med Assoc. 2007. PMID: 17223757 Clinical Trial.
-
Evaluation of efficacy of an inactivated vaccine against bovine respiratory syncytial virus in calves with maternal antibodies.Am J Vet Res. 2004 Apr;65(4):417-21. doi: 10.2460/ajvr.2004.65.417. Am J Vet Res. 2004. PMID: 15077682
-
[Immunobiology of bovine respiratory syncytial virus infections].Tijdschr Diergeneeskd. 1998 Nov 15;123(22):658-62. Tijdschr Diergeneeskd. 1998. PMID: 9836385 Review. Dutch.
-
Bovine respiratory syncytial virus.Vet Clin North Am Food Anim Pract. 2010 Jul;26(2):323-33. doi: 10.1016/j.cvfa.2010.04.010. Vet Clin North Am Food Anim Pract. 2010. PMID: 20619187 Review.
Cited by
-
Productive and physiological responses of feeder cattle supplemented with Yucca schidigera extract during feedlot receiving.J Anim Sci. 2019 Jan 1;97(1):208-219. doi: 10.1093/jas/sky412. J Anim Sci. 2019. PMID: 30351413 Free PMC article.
-
Mucosal Adjuvant Potential of Quillaja saponins and Cross-linked Dextran Microspheres, Co-administered with Liposomes Encapsulated with Tetanus Toxoid.Iran J Pharm Res. 2012 Summer;11(3):723-32. Iran J Pharm Res. 2012. PMID: 24250499 Free PMC article.
-
Association between haptoglobin and IgM levels and the clinical progression of caseous lymphadenitis in sheep.BMC Vet Res. 2013 Dec 13;9:254. doi: 10.1186/1746-6148-9-254. BMC Vet Res. 2013. PMID: 24330714 Free PMC article.
-
Immunization Against Cutaneous Leishmaniasis by Alginate Microspheres Loaded With Autoclaved Leishmania Major (ALM) and Quillaja Saponins.Iran J Pharm Res. 2016 Spring;15(2):573-81. Iran J Pharm Res. 2016. PMID: 27642328 Free PMC article.
-
Intranasal and oral vaccination with protein-based antigens: advantages, challenges and formulation strategies.Protein Cell. 2015 Jul;6(7):480-503. doi: 10.1007/s13238-015-0164-2. Epub 2015 May 6. Protein Cell. 2015. PMID: 25944045 Free PMC article. Review.
References
-
- Van der Poel WH, Brand A, Kramps JA, et al. Respiratory syncytial virus infection in human beings and in cattle. J Infect. 1994;29:215–228. - PubMed
-
- Kimman TG, Westenbrink F. Immunity to human and bovine respiratory syncytial virus. Arch Virol. 1990;112:1–25. - PubMed
-
- Baker JC, Ellis JA, Clark EG. Bovine respiratory syncytial virus. Vet Clin North Am Food Anim Pract. 1997;13:425–454. - PubMed
-
- Bryson DG, McNulty MS, Logan EF, et al. Respiratory syncytial virus pneumonia in young calves: clinical and pathologic findings. Am J Vet Res. 1983;44:1648–1655. - PubMed
-
- Bryson DG. Necropsy findings associated with BRSV pneumonia. Vet Med 1993;88:894,896–899.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
