Targeted inactivation of hepatic Abca1 causes profound hypoalphalipoproteinemia and kidney hypercatabolism of apoA-I
- PMID: 15841208
- PMCID: PMC1074680
- DOI: 10.1172/JCI23915
Targeted inactivation of hepatic Abca1 causes profound hypoalphalipoproteinemia and kidney hypercatabolism of apoA-I
Abstract
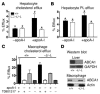
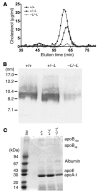
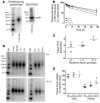
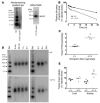
Patients with Tangier disease exhibit extremely low plasma HDL concentrations resulting from mutations in the ATP-binding cassette, sub-family A, member 1 (ABCA1) protein. ABCA1 controls the rate-limiting step in HDL particle assembly by mediating efflux of cholesterol and phospholipid from cells to lipid-free apoA-I, which forms nascent HDL particles. ABCA1 is widely expressed; however, the specific tissues involved in HDL biogenesis are unknown. To determine the role of the liver in HDL biogenesis, we generated mice with targeted deletion of the second nucleotide-binding domain of Abca1 in liver only (Abca1(-L/-L)). Abca1(-L/-L) mice had total plasma and HDL cholesterol concentrations that were 19% and 17% those of wild-type littermates, respectively. In vivo catabolism of HDL apoA-I from wild-type mice or human lipid-free apoA-I was 2-fold higher in Abca1(-L/-L) mice compared with controls due to a 2-fold increase in the catabolism of apoA-I by the kidney, with no change in liver catabolism. We conclude that in chow-fed mice, the liver is the single most important source of plasma HDL. Furthermore, hepatic, but not extrahepatic, Abca1 is critical in maintaining the circulation of mature HDL particles by direct lipidation of hepatic lipid-poor apoA-I, slowing its catabolism by the kidney and prolonging its plasma residence time.
Figures


References
-
- Assmann G, Gotto AM., Jr HDL cholesterol and protective factors in atherosclerosis. Circulation. 2004;109(Suppl. 1):III8–III14. - PubMed
-
- Glomset JA. The plasma lecithin:cholesterol acyltransferase reaction. J. Lipid Res. 1968;9:155–167. - PubMed
-
- Hamilton RL, Moorehouse A, Havel RJ. Isolation and properties of nascent lipoproteins from highly purified rat hepatocytic Golgi fractions. J. Lipid Res. 1991;32:529–543. - PubMed
-
- Hamilton RL, Guo LS, Felker TE, Chao YS, Havel RJ. Nascent high density lipoproteins from liver perfusates of orotic acid-fed rats. J. Lipid Res. 1986;27:967–978. - PubMed
-
- Oram JF, Yokoyama S. Apolipoprotein-mediated removal of cellular cholesterol and phospholipids. J. Lipid Res. 1996;37:2473–2491. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

