T-box genes coordinate regional rates of proliferation and regional specification during cardiogenesis
- PMID: 15843407
- PMCID: PMC5576439
- DOI: 10.1242/dev.01832
T-box genes coordinate regional rates of proliferation and regional specification during cardiogenesis
Abstract
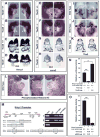
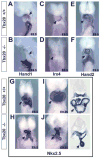
Mutations in T-box genes are the cause of several congenital diseases and are implicated in cancer. Tbx20-null mice exhibit severely hypoplastic hearts and express Tbx2, which is normally restricted to outflow tract and atrioventricular canal, throughout the heart. Tbx20 mutant hearts closely resemble those seen in mice overexpressing Tbx2 in myocardium, suggesting that upregulation of Tbx2 can largely account for the cardiac phenotype in Tbx20-null mice. We provide evidence that Tbx2 is a direct target for repression by Tbx20 in developing heart. We have also found that Tbx2 directly binds to the Nmyc1 promoter in developing heart, and can repress expression of the Nmyc1 promoter in transient transfection studies. Repression of Nmyc1 (N-myc) by aberrantly regulated Tbx2 can account in part for the observed cardiac hypoplasia in Tbx20 mutants. Nmyc1 is required for growth and development of multiple organs, including the heart, and overexpression of Nmyc1 is associated with childhood tumors. Despite its clinical relevance, the factors that regulate Nmyc1 expression during development are unknown. Our data present a paradigm by which T-box proteins regulate regional differences in Nmyc1 expression and proliferation to effect organ morphogenesis. We present a model whereby Tbx2 directly represses Nmyc1 in outflow tract and atrioventricular canal of the developing heart, resulting in relatively low proliferation. In chamber myocardium, Tbx20 represses Tbx2, preventing repression of Nmyc1 and resulting in relatively high proliferation. In addition to its role in regulating regional proliferation, we have found that Tbx20 regulates expression of a number of genes that specify regional identity within the heart, thereby coordinating these two important aspects of organ development.
Figures






References
-
- Brummelkamp TR, Kortlever RM, Lingbeek M, Trettel F, MacDonald ME, van Lohuizen M, Bernards R. TBX-3, the gene mutated in Ulnar-Mammary Syndrome, is a negative regulator of p19ARF and inhibits senescence. J Biol Chem. 2002;277:6567–6572. - PubMed
-
- Bruneau BG, Bao ZZ, Tanaka M, Schott JJ, Izumo S, Cepko CL, Seidman JG, Seidman CE. Cardiac expression of the ventricle-specific homeobox gene Irx4 is modulated by Nkx2-5 and dHand. Dev Biol. 2000;217:266–277. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

