The Arabidopsis AtRaptor genes are essential for post-embryonic plant growth
- PMID: 15845148
- PMCID: PMC1131892
- DOI: 10.1186/1741-7007-3-12
The Arabidopsis AtRaptor genes are essential for post-embryonic plant growth
Abstract
Background: Flowering plant development is wholly reliant on growth from meristems, which contain totipotent cells that give rise to all post-embryonic organs in the plant. Plants are uniquely able to alter their development throughout their lifespan through the generation of new organs in response to external signals. To identify genes that regulate meristem-based growth, we considered homologues of Raptor proteins, which regulate cell growth in response to nutrients in yeast and metazoans as part of a signaling complex with the target of rapamycin (TOR) kinase.
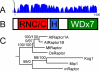
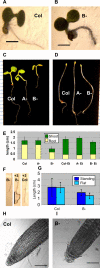
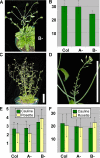
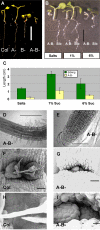
Results: We identified AtRaptor1A and AtRaptor1B, two loci predicted to encode Raptor proteins in Arabidopsis. Disruption of AtRaptor1B yields plants with a wide range of developmental defects: roots are thick and grow slowly, leaf initiation and bolting are delayed and the shoot inflorescence shows reduced apical dominance. AtRaptor1A AtRaptor1B double mutants show normal embryonic development but are unable to maintain post-embryonic meristem-driven growth. AtRaptor transcripts accumulate in dividing and expanding cells and tissues.
Conclusion: The data implicate the TOR signaling pathway, a major regulator of cell growth in yeast and metazoans, in the maintenance of growth from the shoot apical meristem in plants. These results provide insights into the ways in which TOR/Raptor signaling has been adapted to regulate plant growth and development, and indicate that in plants, as in other eukaryotes, there is some Raptor-independent TOR activity.
Figures
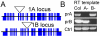


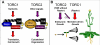
References
-
- Thomas G, Sabatini D, Hall M. Current Topics in Microbiology and Immunology 279. New York: Springer-Verlag; 2004. TOR Target of Rapamycin.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

