Mutagenesis by reversible promoter insertion to study the activation of NF-kappaB
- PMID: 15851657
- PMCID: PMC1088393
- DOI: 10.1073/pnas.0502463102
Mutagenesis by reversible promoter insertion to study the activation of NF-kappaB
Abstract
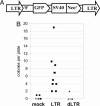
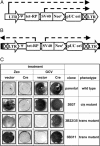
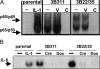
Genetic dissection of signaling pathways in mammalian cells involves screening or selecting phenotypic mutants obtained by a variety of techniques. Limitations in current methods include inadequate genome coverage and difficulty in validating the link between mutation and phenotype. We describe an improved method for insertional mutagenesis with retroviral vectors and show that the ability to induce mutations increases greatly if a randomly inserted promoter directs transcription into the host DNA. The mutant phenotype is due to the expression of a hybrid transcript derived from the vector and the insertion site. Because other alleles of the affected gene remain intact, the phenotype is dominant, but is reversible by inactivating the promoter, for example, by site-specific recombination. Importantly, in mutant clones with multiple inserts, limited excision yields progeny with different patterns of inserts remaining. Characterizing these progeny allows the mutant phenotype to be associated with a specific target gene. Relative simplicity and robust target validation make the method suitable for a broad range of applications. We have used this technique to search for proteins that regulate NF-kappaB-dependent signaling in human cells. Two validated targets are the relA gene, which codes for the NF-kappaB p65 subunit, and the NF-kappaB regulator act1. Overexpression of the corresponding proteins, caused by insertion of a promoter into the first intron of each gene, leads to NF-kappaB-dependent secretion of factors that activate NF-kappaB through cell-surface receptors, establishing an autocrine loop.
Figures


Similar articles
-
Use of forward genetics to discover novel regulators of NF-kappaB.Cold Spring Harb Perspect Biol. 2010 Jun;2(6):a001966. doi: 10.1101/cshperspect.a001966. Epub 2009 Dec 23. Cold Spring Harb Perspect Biol. 2010. PMID: 20516132 Free PMC article. Review.
-
Validation-based insertional mutagenesis identifies lysine demethylase FBXL11 as a negative regulator of NFkappaB.Proc Natl Acad Sci U S A. 2009 Sep 22;106(38):16339-44. doi: 10.1073/pnas.0908560106. Epub 2009 Sep 1. Proc Natl Acad Sci U S A. 2009. PMID: 19805303 Free PMC article.
-
Bidirectional regulation of nuclear factor-κB and mammalian target of rapamycin signaling functionally links Bnip3 gene repression and cell survival of ventricular myocytes.Circ Heart Fail. 2013 Mar;6(2):335-43. doi: 10.1161/CIRCHEARTFAILURE.112.000061. Epub 2013 Feb 8. Circ Heart Fail. 2013. PMID: 23395931
-
Participation of the E3-ligase TRIM13 in NF-κB p65 activation and NFAT-dependent activation of c-Rel upon T-cell receptor engagement.Int J Biochem Cell Biol. 2014 Sep;54:217-22. doi: 10.1016/j.biocel.2014.07.012. Epub 2014 Aug 1. Int J Biochem Cell Biol. 2014. PMID: 25088585
-
Thioredoxin 2 Is a Novel E2-Interacting Protein That Inhibits the Replication of Classical Swine Fever Virus.J Virol. 2015 Aug;89(16):8510-24. doi: 10.1128/JVI.00429-15. Epub 2015 Jun 3. J Virol. 2015. PMID: 26041303 Free PMC article.
Cited by
-
Use of forward genetics to discover novel regulators of NF-kappaB.Cold Spring Harb Perspect Biol. 2010 Jun;2(6):a001966. doi: 10.1101/cshperspect.a001966. Epub 2009 Dec 23. Cold Spring Harb Perspect Biol. 2010. PMID: 20516132 Free PMC article. Review.
-
How cells respond to interferons revisited: from early history to current complexity.Cytokine Growth Factor Rev. 2007 Oct-Dec;18(5-6):419-23. doi: 10.1016/j.cytogfr.2007.06.013. Epub 2007 Aug 1. Cytokine Growth Factor Rev. 2007. PMID: 17683974 Free PMC article. Review.
-
Protein kinase A type II-α regulatory subunit regulates the response of prostate cancer cells to taxane treatment.Cell Cycle. 2014;13(20):3292-301. doi: 10.4161/15384101.2014.949501. Cell Cycle. 2014. PMID: 25485509 Free PMC article.
-
Long-distance effects of insertional mutagenesis.PLoS One. 2011 Jan 5;6(1):e15832. doi: 10.1371/journal.pone.0015832. PLoS One. 2011. PMID: 21246045 Free PMC article.
-
The role of polo-like kinase 3 in the response of BRAF-mutant cells to targeted anticancer therapies.Mol Carcinog. 2020 Jan;59(1):5-14. doi: 10.1002/mc.23123. Epub 2019 Sep 30. Mol Carcinog. 2020. PMID: 31571292 Free PMC article.
References
-
- Stark, G. R. & Gudkov, A. V. (1999) Hum. Mol. Genet. 8, 1925-1938. - PubMed
-
- Kandel, E. S. & Stark, G. R. (2003) in Signal Transducers and Activators of Transcription (STATs): Activation and Biology, eds. Sehgal, P. B., Levy, D. E. & Hirano, T. (Kluwer, Dodrecht, The Netherlands), pp. 299-309.
-
- Li, L. & Cohen, S. N. (1996) Cell 85, 319-329. - PubMed
-
- Liu, D., Yang, X., Yang, D. & Songyang, Z. (2000) Oncogene 19, 5964-5972. - PubMed
-
- Lin, S. Y. & Elledge, S. J. (2003) Cell 113, 881-889. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

