Spontaneous corneal hem- and lymphangiogenesis in mice with destrin-mutation depend on VEGFR3 signaling
- PMID: 15855638
- PMCID: PMC1606392
- DOI: 10.1016/S0002-9440(10)62355-3
Spontaneous corneal hem- and lymphangiogenesis in mice with destrin-mutation depend on VEGFR3 signaling
Abstract
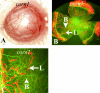
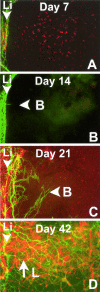
Lymphangiogenesis, the formation of new lymphatic vessels, is important for tumor metastasis and induction of immunity to peripheral antigens including organ transplants. We herein describe a novel mouse model of spontaneous, secondary lymphangiogenesis in the normally avascular cornea. corn1 mice, which suffer from a deletion in the gene encoding the cytoskeletal protein destrin, develop hemangiogenesis as well as spontaneous outgrowth of LYVE-1+++/CD31+ lymphatic vessels into the cornea starting at age 4 weeks. Corneal lymphangiogenesis is delayed in onset, is less intense, and regresses earlier compared with hemangiogenesis. Moreover, the lymphangiogenesis is preceded only by a mild recruitment of CD45+ inflammatory cells into the cornea. In contrast to mice with inflammation-induced hem- and lymphangiogenesis, corn1 mice do not develop breakdown of the blood-aqueous barrier. Finally, in this novel mouse model, a blocking anti-VEGFR3 antibody significantly inhibited not only lymph- but also hemangiogenesis. In summary, destrin deletion has differential effects on spontaneous hem- and lymphangiogenesis in the normally avascular cornea and represents a novel mouse model to study the mechanisms of lymphangiogenesis and to test the antihem- and antilymphangiogenic properties of known or new antiangiogenic agents.
Figures
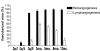




References
-
- Alitalo K, Carmeliet P. Molecular mechanisms of lymphangiogenesis in health and disease. Cancer Cell. 2002;1:219–227. - PubMed
-
- Cursiefen C, Chen L, Dana MR, Streilein JW. Corneal lymphangiogenesis: evidence, mechanisms, and implications for corneal transplant immunology. Cornea. 2003;22:273–281. - PubMed
-
- He Y, Kozaki K, Karpanen T, Koshikawa K, Yla-Herttuala S, Takahashi T, Alitalo K. Suppression of tumor lymphangiogenesis and lymph node metastasis by blocking vascular endothelial growth factor receptor 3 signaling. J Natl Cancer Inst. 2002;94:819–825. - PubMed
-
- Skobe M, Hawighorst T, Jackson DG, Prevo R, Janes L, Velasco P, Riccardi L, Alitalo K, Claffey K, Detmar M. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat Med. 2001;7:192–198. - PubMed
-
- Cursiefen C, Chen L, Yiu L, Maryuama K, Jackson D, Dana MR, Wiegand S, Streilein JW. Inhibition of hem- and lymphangiogenesis after normal-risk keratoplasty by neutralizing VEGF improves corneal transplant survival. Invest Ophthalmol Vis Sci. 2004;45:2666–2673. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

