Respiration and growth of Shewanella oneidensis MR-1 using vanadate as the sole electron acceptor
- PMID: 15866913
- PMCID: PMC1111990
- DOI: 10.1128/JB.187.10.3293-3301.2005
Respiration and growth of Shewanella oneidensis MR-1 using vanadate as the sole electron acceptor
Abstract
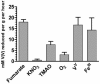
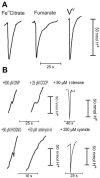
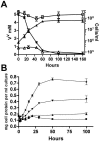
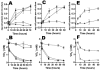
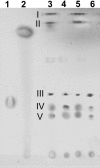
Shewanella oneidensis MR-1 is a free-living gram-negative gamma-proteobacterium that is able to use a large number of oxidizing molecules, including fumarate, nitrate, dimethyl sulfoxide, trimethylamine N-oxide, nitrite, and insoluble iron and manganese oxides, to drive anaerobic respiration. Here we show that S. oneidensis MR-1 is able to grow on vanadate as the sole electron acceptor. Oxidant pulse experiments demonstrated that proton translocation across the cytoplasmic membrane occurs during vanadate reduction. Proton translocation is abolished in the presence of protonophores and the inhibitors 2-heptyl-4-hydroxyquinoline N-oxide and antimycin A. Redox difference spectra indicated the involvement of membrane-bound menaquinone and cytochromes c, which was confirmed by transposon mutagenesis and screening for a vanadate reduction-deficient phenotype. Two mutants which are deficient in menaquinone synthesis were isolated. Another mutant with disruption in the cytochrome c maturation gene ccmA was unable to produce any cytochrome c and to grow on vanadate. This phenotype could be restored by complementation with the pEC86 plasmid expressing ccm genes from Escherichia coli. To our knowledge, this is the first report of E. coli ccm genes being functional in another organism. Analysis of an mtrB-deficient mutant confirmed the results of a previous paper indicating that OmcB may function as a vanadate reductase or may be part of a vanadate reductase complex.
Figures
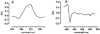

Similar articles
-
A kinetic approach to the dependence of dissimilatory metal reduction by Shewanella oneidensis MR-1 on the outer membrane cytochromes c OmcA and OmcB.FEBS J. 2007 Jul;274(14):3728-3738. doi: 10.1111/j.1742-4658.2007.05907.x. Epub 2007 Jul 2. FEBS J. 2007. PMID: 17608722
-
Shewanella oneidensis MR-1 restores menaquinone synthesis to a menaquinone-negative mutant.Appl Environ Microbiol. 2004 Sep;70(9):5415-25. doi: 10.1128/AEM.70.9.5415-5425.2004. Appl Environ Microbiol. 2004. PMID: 15345428 Free PMC article.
-
Regulation of Gene Expression in Shewanella oneidensis MR-1 during Electron Acceptor Limitation and Bacterial Nanowire Formation.Appl Environ Microbiol. 2016 Aug 15;82(17):5428-43. doi: 10.1128/AEM.01615-16. Print 2016 Sep 1. Appl Environ Microbiol. 2016. PMID: 27342561 Free PMC article.
-
Respiration of metal (hydr)oxides by Shewanella and Geobacter: a key role for multihaem c-type cytochromes.Mol Microbiol. 2007 Jul;65(1):12-20. doi: 10.1111/j.1365-2958.2007.05783.x. Mol Microbiol. 2007. PMID: 17581116 Free PMC article. Review.
-
Structural and functional studies of multiheme cytochromes C involved in extracellular electron transport in bacterial dissimilatory metal reduction.Biochemistry (Mosc). 2014 Dec;79(13):1584-601. doi: 10.1134/S0006297914130094. Biochemistry (Mosc). 2014. PMID: 25749166 Review.
Cited by
-
Application of DFT methods to the study of the coordination environment of the VO2+ ion in V proteins.J Biol Inorg Chem. 2012 Jun;17(5):773-90. doi: 10.1007/s00775-012-0895-y. Epub 2012 Apr 15. J Biol Inorg Chem. 2012. PMID: 22526563
-
Extracellular electron transport-mediated Fe(III) reduction by a community of alkaliphilic bacteria that use flavins as electron shuttles.Appl Environ Microbiol. 2014 Jan;80(1):128-37. doi: 10.1128/AEM.02282-13. Epub 2013 Oct 18. Appl Environ Microbiol. 2014. PMID: 24141133 Free PMC article.
-
Mechanism and Consequences of anaerobic respiration of cobalt by Shewanella oneidensis strain MR-1.Appl Environ Microbiol. 2008 Nov;74(22):6880-6. doi: 10.1128/AEM.00840-08. Epub 2008 Oct 3. Appl Environ Microbiol. 2008. PMID: 18836009 Free PMC article.
-
Menaquinone and iron are essential for complex colony development in Bacillus subtilis.PLoS One. 2013 Nov 4;8(11):e79488. doi: 10.1371/journal.pone.0079488. eCollection 2013. PLoS One. 2013. PMID: 24223955 Free PMC article.
-
Green Synthesis of Vanadium Dioxide Nanoparticles by Shewanella sp. Strain HN-41.J Microbiol Biotechnol. 2025 May 26;35:e2502051. doi: 10.4014/jmb.2502.02051. J Microbiol Biotechnol. 2025. PMID: 40443232 Free PMC article.
References
-
- Ahuja, U., and L. Töny-Meyer. 2003. Dynamic features of a heme delivery system for cytochrome c maturation. J. Biol. Chem. 278:52061-52070. - PubMed
-
- Akagawamatsushita, M., T. Itoh, Y. Katayama, H. Kuraishi, and K. Yamasato. 1992. Isoprenoid quinone composition of some marine Alteromonas, Marinomonas, Deleya, Pseudomonas and Shewanella species. J. Gen. Microbiol. 138:2275-2281.
-
- Armstrong, P. B., W. B. Lyons, and H. E. Gaudette. 1979. Application of formaldoxime colorimetric method for the determination of manganese in the pore water of anoxic estuarine sediments. Estuaries 2:198-201.
-
- Arslan, E., H. Schulz, R. Zufferey, P. Kunzle, and L. Thöny-Meyer. 1998. Overproduction of the Bradyrhizobium japonicum c-type cytochrome subunits of the cbb3 oxidase in Escherichia coli. Biochem. Biophys. Res. Commun. 251:744-747. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

