A small conserved surface in SUMO is the critical structural determinant of its transcriptional inhibitory properties
- PMID: 15870296
- PMCID: PMC1087732
- DOI: 10.1128/MCB.25.10.4272-4282.2005
A small conserved surface in SUMO is the critical structural determinant of its transcriptional inhibitory properties
Abstract
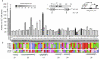
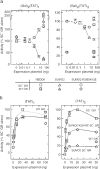
Small ubiquitin-like modifier (SUMO) modification of sequence-specific transcription factors has profound regulatory consequences. By providing an intrinsic inhibitory function, SUMO isoforms can suppress transcriptional activation, particularly at promoters harboring multiple response elements. Through a comprehensive structure-function analysis, we have identified a single critical sector along the second beta sheet and the following alpha helix of SUMO2. This distinct surface is defined by four basic residues (K33, K35, K42, R50) that surround a shallow pocket lined by aliphatic (V30, I34) and polar (T38) residues. Substitutions within this area specifically and dramatically affected the ability of both SUMO2 and SUMO1 to inhibit transcription and revealed that the positively charged nature of the key basic residues is the main feature responsible for their functional role. This highly conserved surface accounts for the inhibitory properties of SUMO on multiple transcription factors and promoter contexts and likely defines the interaction surface for the corepressors that mediate the inhibitory properties of SUMO.
Figures







References
-
- Abdel-Hafiz, H., G. S. Takimoto, L. Tung, and K. B. Horwitz. 2002. The inhibitory function in human progesterone receptor N termini binds SUMO-1 protein to regulate autoinhibition and transrepression. J. Biol. Chem. 277:33950-33956. - PubMed
-
- Bailey, D., and P. O'Hare. 2004. Characterization of the localization and proteolytic activity of the SUMO-specific protease, SENP1. J. Biol. Chem. 279:692-703. - PubMed
-
- Bies, J., J. Markus, and L. Wolff. 2002. Covalent attachment of the SUMO-1 protein to the negative regulatory domain of the c-Myb transcription factor modifies its stability and transactivation capacity. J. Biol. Chem. 277:8999-9009. - PubMed
-
- Bohren, K. M., V. Nadkarni, J. H. Song, K. H. Gabbay, and D. Owerbach. 2004. A M55V Polymorphism in a novel SUMO gene (SUMO-4) differentially activates heat shock transcription factors and is associated with susceptibility to type I diabetes mellitus. J. Biol. Chem. 279:27233-27238. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
