The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory T cells in peripheral tolerance
- PMID: 15883173
- PMCID: PMC2212925
- DOI: 10.1084/jem.20042307
The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory T cells in peripheral tolerance
Abstract
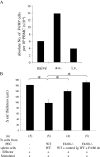
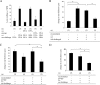
We show that the mouse macrophage-restricted F4/80 protein is not required for the development and distribution of tissue macrophages but is involved in the generation of antigen-specific efferent regulatory T (T reg) cells that suppress antigen-specific immunity. In the in vivo anterior chamber (a.c.)-associated immune deviation (ACAID) model of peripheral tolerance, a.c. inoculation of antigen into F4/80(-/-) mice was unable to induce efferent T reg cells and suppress delayed-type hypersensitivity (DTH) responses. Moreover, the use of anti-F4/80 mAb and F4/80(-/-) APCs in an in vitro ACAID model showed that all APC cells in the culture must be able to express F4/80 protein if efferent T reg cells were to be generated. In a low-dose oral tolerance model, WT but not F4/80(-/-) mice generated an efferent CD8(+) T reg cell population that suppressed an antigen-specific DTH response. Peripheral tolerance was restored in F4/80(-/-) mice by adoptive transfer of F4/80(+) APCs in both peripheral tolerance models, indicating a central role for the F4/80 molecule in the generation of efferent CD8(+) T reg cells.
Figures




References
-
- Gordon, S. 1998. The role of the macrophage in immune regulation. Res. Immunol. 149:685–688. - PubMed
-
- Stahl, P.D. 1992. The mannose receptor and other macrophage lectins. Curr. Opin. Immunol. 4:49–52. - PubMed
-
- Platt, N., R.P. da Silva, and S. Gordon. 1999. Class A scavenger receptors and the phagocytosis of apoptotic cells. Immunol. Lett. 65:15–19. - PubMed
-
- Austyn, J.M., and S. Gordon. 1981. F4/80, a monoclonal antibody directed specifically against the mouse macrophage. Eur. J. Immunol. 11:805–815. - PubMed
-
- Morris, L., C.F. Graham, and S. Gordon. 1991. Macrophages in haemopoietic and other tissues of the developing mouse detected by the monoclonal antibody F4/80. Development. 112:517–526. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

