The activating NKG2D receptor assembles in the membrane with two signaling dimers into a hexameric structure
- PMID: 15894612
- PMCID: PMC1140444
- DOI: 10.1073/pnas.0502439102
The activating NKG2D receptor assembles in the membrane with two signaling dimers into a hexameric structure
Abstract
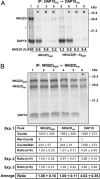
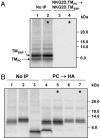
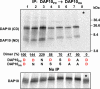
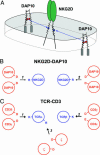
The activating NKG2D receptor plays a critical role in innate and adaptive immune responses by natural killer cells and subpopulations of T cells. The human receptor assembles with the DAP10 signaling dimer, and it is thought that one NKG2D homodimer pairs with a single DAP10 dimer by formation of two salt bridges between charged transmembrane (TM) residues. However, direct stoichiometry measurements demonstrated that one NKG2D homodimer assembles with four DAP10 chains. Selective mutation of one of the basic TM residues of NKG2D resulted in loss of two DAP10 chains, indicating that each TM arginine serves as an interaction site for a DAP10 dimer. Assembly of the hexameric structure was cooperative because this mutation also significantly reduced NKG2D dimerization. A monomeric NKG2D TM peptide was sufficient for assembly with a DAP10 dimer, indicating that the interaction between these proteins occurs in the membrane environment. Formation of a three-helix interface among the TM domains involved ionizable residues from all three chains, the TM arginine of NKG2D and both TM aspartic acids of the DAP10 dimer. The organization of the TM domains thus shows similarities to the T cell antigen receptor-CD3 complex, in particular to the six-chain assembly intermediate between T cell antigen receptor and the CD3delta epsilon and CD3gamma epsilon dimers. Binding of a single ligand can thus result in phosphorylation of four DAP10 chains, which may be relevant for the sensitivity of NKG2D receptor signaling, in particular in situations of low ligand density.
Figures



References
-
- Bauer, S., Groh, V., Wu, J., Steinle, A., Phillips, J. H., Lanier, L. L. & Spies, T. (1999) Science 285, 727–729. - PubMed
-
- Wu, J., Song, Y., Bakker, A. B., Bauer, S., Spies, T., Lanier, L. L. & Phillips, J. H. (1999) Science 285, 730–732. - PubMed
-
- Cerwenka, A., Bakker, A. B., McClanahan, T., Wagner, J., Wu, J., Phillips, J. H. & Lanier, L. L. (2000) Immunity 12, 721–727. - PubMed
-
- Cosman, D., Mullberg, J., Sutherland, C. L., Chin, W., Armitage, R., Fanslow, W., Kubin, M. & Chalupny, N. J. (2001) Immunity 14, 123–133. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

