Developmental changes in AMPA and kainate receptor-mediated quantal transmission at thalamocortical synapses in the barrel cortex
- PMID: 15917466
- PMCID: PMC6724821
- DOI: 10.1523/JNEUROSCI.0827-05.2005
Developmental changes in AMPA and kainate receptor-mediated quantal transmission at thalamocortical synapses in the barrel cortex
Erratum in
- J Neurosci. 2005 Jun 8;25(23):5666
Abstract
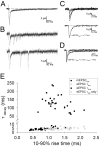
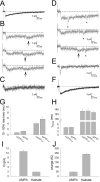
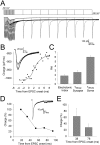
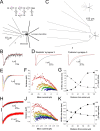
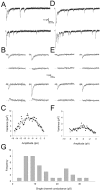
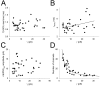
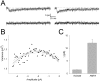
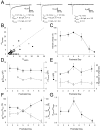
During the first week of life, there is a shift from kainate to AMPA receptor-mediated thalamocortical transmission in layer IV barrel cortex. However, the mechanisms underlying this change and the differential properties of AMPA and kainate receptor-mediated transmission remain essentially unexplored. To investigate this, we studied the quantal properties of AMPA and kainate receptor-mediated transmission using strontium-evoked miniature EPSCs. AMPA and kainate receptor-mediated transmission exhibited very different quantal properties but were never coactivated by a single quantum of transmitter, indicating complete segregation to different synapses within the thalamocortical input. Nonstationary fluctuation analysis showed that synaptic AMPA receptors exhibited a range of single-channel conductance (gamma) and a strong negative correlation between gamma and functional channel number, indicating that these two parameters are reciprocally regulated at thalamocortical synapses. We obtained the first estimate of gamma for synaptic kainate receptors (<2 pS), and this primarily accounted for the small quantal size of kainate receptor-mediated transmission. Developmentally, the quantal contribution to transmission of AMPA receptors increased and that of kainate receptors decreased. No changes in AMPA or kainate quantal amplitude or in AMPA receptor gamma were observed, demonstrating that the developmental change was attributable to a decrease in the number of kainate synapses and an increase in the number of AMPA synapses contributing to transmission. Therefore, we demonstrate fundamental differences in the quantal properties for these two types of synapse. Thus, the developmental switch in transmission will dramatically alter information transfer at thalamocortical inputs to layer IV.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
