A single course of anti-CD3 monoclonal antibody hOKT3gamma1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes
- PMID: 15919798
- PMCID: PMC5315015
- DOI: 10.2337/diabetes.54.6.1763
A single course of anti-CD3 monoclonal antibody hOKT3gamma1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes
Abstract
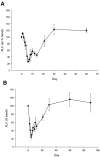
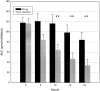
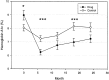
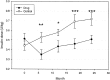
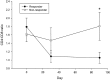
Despite advances in understanding autoimmune diabetes in animal models, there has been little progress in altering the natural course of the human disease, which involves progression to insulin deficiency. Studies with immunosuppressive agents have shown short-term effectiveness, but they have not induced tolerance, and continuous treatment is needed. We studied the effects of hOKT3gamma1(Ala-Ala), a humanized Fc mutated anti-CD3 monoclonal antibody, on the progression of type 1 diabetes in patients with recent-onset disease in a randomized controlled trial. In general, the drug was well tolerated. A single course of treatment, within the first 6 weeks after diagnosis, preserved C-peptide responses to a mixed meal for 1 year after diagnosis (97 +/- 9.6% of response at study entry in drug-treated patients vs. 53 +/- 7.6% in control subjects, P < 0.01), with significant improvement in C-peptide responses to a mixed meal even 2 years after treatment (P < 0.02). The improved C-peptide responses were accompanied by reduced HbA(1c) and insulin requirements. Clinical responses to drug treatment were predicted by an increase in the relative number of CD8(+) T-cells in the peripheral blood after the lymphocyte count recovered 2 weeks after the last dose of drug. We conclude that treatment with the anti-CD3 monoclonal antibody hOKT3gamma1(Ala-Ala) results in improved C-peptide responses and clinical parameters in type 1 diabetes for at least 2 years in the absence of continued immunosuppressive medications.
Figures

References
-
- Stiller CR, Dupre J, Gent M, Jenner MR, Keown PA, Laupacis A, Martell R, Rodger NW, von Graffenried B, Wolfe BM: Effects of cyclosporine immunosuppression in insulin-dependent diabetes mellitus of recent onset. Science 223: 1362–1367, 1984 - PubMed
-
- Skyler JS, Lorenz TJ, Schwartz S, Eisenbarth GS, Einhorn D, Palmer JP, Marks JB, Greenbaum C, Saria EA, Byers V: Effects of an anti-CD5 immunoconjugate (CD5-plus) in recent onset type I diabetes mellitus: a preliminary investigation. The CD5 Diabetes Project Team. J Diabetes Complications 7: 224–232, 1993 - PubMed
-
- Skyler JS, Rabinovitch A: Cyclosporine in recent onset type I diabetes mellitus: effects on islet beta cell function. Miami Cyclosporine Diabetes Study Group. J Diabetes Complications 6: 77–88, 1992 - PubMed
-
- Bougneres PF, Carel JC, Castano L, Boitard C, Gardin JP, Landais P, Hors J, Mihatsch MJ, Paillard M, Chaussain JL, et al.: Factors associated with early remission of type I diabetes in children treated with cyclosporine. N Engl J Med 318: 663–670, 1988 - PubMed
-
- Buckingham BA, Sandborg CI: A randomized trial of methotrexate in newly diagnosed patients with type 1 diabetes mellitus. Clin Immunol 96: 86–90, 2000 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

