Integrin alpha(v)beta8-mediated activation of transforming growth factor-beta by perivascular astrocytes: an angiogenic control switch
- PMID: 15920172
- PMCID: PMC1602409
- DOI: 10.1016/s0002-9440(10)62497-2
Integrin alpha(v)beta8-mediated activation of transforming growth factor-beta by perivascular astrocytes: an angiogenic control switch
Abstract
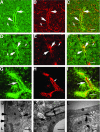
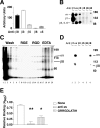
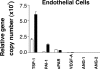
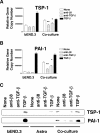
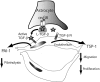
Brain hemorrhage is a severe complication of both neoplastic and nonneoplastic brain disease. Mice deficient in the alpha(v)beta8 integrin display defective brain vessel formation resulting in hemorrhage and perinatal death, but the mechanism of brain hemorrhage is unknown. Because the alpha(v)beta8 integrin is expressed by astrocytes and not expressed by endothelium, paracrine interactions between astrocytes and endothelial cells could contribute to the maintenance of brain vessel integrity. We have investigated the mechanisms underlying astrocytic-endothelial paracrine signaling and have found that integrin-mediated activation of transforming growth factor (TGF)-beta by astrocytes influences endothelial cell function. Thus, we identified the integrin alpha(v)beta8 in human perivascular glial cell processes surrounding developing blood vessels. Human astrocytic alpha(v)beta8 was a major cell surface receptor for latent TGF-beta, and alpha(v)beta8-dependent activation of TGF-beta was the major mechanism of TGF-beta activation in primary cultures of astrocytes or freshly dissociated fetal brain cells. This activation of TGF-beta was sufficient to inhibit endothelial migration in fibrin gels and to alter expression of genes affecting proteolytic and angiogenic pathways. Taken together, our data suggest that astrocytic alpha(v)beta8 acts as a central regulator of brain vessel homeostasis through regulation of TGF-beta activation and expression of TGF-beta-responsive genes that promote vessel differentiation and stabilization, most notably plasminogen activator inhibitor-1 and thrombospondin-1.
Figures
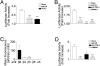

References
-
- Ballabh P, Braun A, Nedergaard M. The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol Dis. 2004;16:1–13. - PubMed
-
- Pardridge WM. Blood-brain barrier biology and methodology. J Neurovirol. 1999;5:556–569. - PubMed
-
- Martin JS, Dickson MC, Cousins FM, Kulkarni AB, Karlsson S, Akhurst RJ. Analysis of homozygous TGF beta 1 null mouse embryos demonstrates defects in yolk sac vasculogenesis and hematopoiesis. Ann NY Acad Sci. 1995;752:300–308. - PubMed
-
- van den Driesche S, Mummery CL, Westermann CJ. Hereditary hemorrhagic telangiectasia: an update on transforming growth factor beta signaling in vasculogenesis and angiogenesis. Cardiovasc Res. 2003;58:20–31. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

