The limit of photoreceptor sensitivity: molecular mechanisms of dark noise in retinal cones
- PMID: 15928405
- PMCID: PMC2234084
- DOI: 10.1085/jgp.200509277
The limit of photoreceptor sensitivity: molecular mechanisms of dark noise in retinal cones
Erratum in
- J Gen Physiol. 2006 Feb;127(2):219-20
Abstract
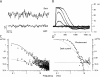
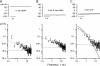
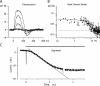
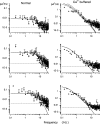
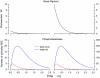
Detection threshold in cone photoreceptors requires the simultaneous absorption of several photons because single photon photocurrent is small in amplitude and does not exceed intrinsic fluctuations in the outer segment dark current (dark noise). To understand the mechanisms that limit light sensitivity, we characterized the molecular origin of dark noise in intact, isolated bass single cones. Dark noise is caused by continuous fluctuations in the cytoplasmic concentrations of both cGMP and Ca(2+) that arise from the activity in darkness of both guanylate cyclase (GC), the enzyme that synthesizes cGMP, and phosphodiesterase (PDE), the enzyme that hydrolyzes it. In cones loaded with high concentration Ca(2+) buffering agents, we demonstrate that variation in cGMP levels arise from fluctuations in the mean PDE enzymatic activity. The rates of PDE activation and inactivation determine the quantitative characteristics of the dark noise power density spectrum. We developed a mathematical model based on the dynamics of PDE activity that accurately predicts this power spectrum. Analysis of the experimental data with the theoretical model allows us to determine the rates of PDE activation and deactivation in the intact photoreceptor. In fish cones, the mean lifetime of active PDE at room temperature is approximately 55 ms. In nonmammalian rods, in contrast, active PDE lifetime is approximately 555 ms. This remarkable difference helps explain why cones are noisier than rods and why cone photocurrents are smaller in peak amplitude and faster in time course than those in rods. Both these features make cones less light sensitive than rods.
Figures


References
-
- Beavo, J.A. 1995. Cyclic nucleotide phosphodiesterases: functional implications of multiple isoforms. Physiol. Rev. 75:725–748. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

