Deafness-related decreases in glycine-immunoreactive labeling in the rat cochlear nucleus
- PMID: 15929063
- PMCID: PMC4455948
- DOI: 10.1002/jnr.20542
Deafness-related decreases in glycine-immunoreactive labeling in the rat cochlear nucleus
Abstract
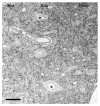
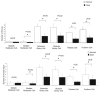
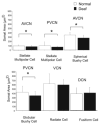
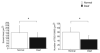
There is increasing evidence of activity-related plasticity in auditory pathways. The present study examined the effects of decreased activity on immunolocalization of the inhibitory neurotransmitter glycine in the cochlear nucleus of the rat after bilateral cochlear ablation. Specifically, glycine-immunoreactive puncta adjacent to somatic profiles were compared in normal hearing animals and animals deafened for 14 days. The number of glycine-immunoreactive puncta surrounding somatic profiles of spherical and globular bushy cells, glycine-immunoreactive type I stellate multipolar cells, radiate neurons (type II stellate multipolar cells), and fusiform cells decreased significantly. In addition, the number of glycine immunopositive tuberculoventral (vertical or corn) cells in the deep layer of the dorsal cochlear nucleus also decreased significantly. These results suggest that decreased inhibition reported in cochlear nucleus after deafness may be due to decreases in glycine.
Copyright 2005 Wiley-Liss, Inc.
Figures

References
-
- Adams JC, Mugnaini E. Immunocytochemical evidence for inhibitory and disinhibitory circuits in the superior olive. Hear Res. 1990;49:281–298. - PubMed
-
- Altschuler RA, Betz H, Parakkal MH, Reeks KA, Wenthold RJ. Identification of glycinergic synapses in the cochlear nucleus through immunocytochemical localization of the postsynaptic receptor. Brain Res. 1986;369:316–320. - PubMed
-
- Babalian AL, Jacomme AV, Doucet JR, Ryugo DK, Rouiller EM. Commissural glycinergic inhibition of bushy and stellate cells in the anteroventral cochlear nucleus. Neuroreport. 2002;13:555–558. - PubMed
-
- Bauer CA, Brozoski TJ, Holder TM, Caspary DM. Effects of chronic salicylate on GABAergic activity in rat inferior colliculus. Hear Res. 2000;147:175–182. - PubMed
-
- Bendiske J, Morest DK. Plastic changes in synaptic vesicle distribution in the cochlear nucleus following noise exposure in mice. Assoc Res Otolaryngol Abs. 2004:300.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

