Caveolin-1 gene knockout impairs nitrergic function in mouse small intestine
- PMID: 15937515
- PMCID: PMC1576236
- DOI: 10.1038/sj.bjp.0706289
Caveolin-1 gene knockout impairs nitrergic function in mouse small intestine
Abstract
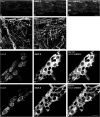
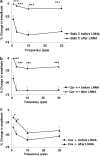
Caveolin-1 is a plasma membrane-associated protein that is responsible for caveolae formation. It plays an important role in the regulation of the function of different signaling molecules, among which are the different isoforms of nitric oxide synthase (NOS). Nitric oxide (NO) is known to be an important inhibitory mediator in the mouse gut. Caveolin-1 knockout mice (Cav1(-/-)) were used to examine the effect of caveolin-1 absence on the NO function in the mouse small intestine (ileum and jejunum) compared to their genetic controls and BALB/c controls. Immunohistochemical staining showed loss of caveolin-1 and NOS in the jejunal smooth muscles and myenteric plexus interstitial cells of Cajal (ICC) of Cav1(-/-) mice; however, nNOS immunoreactive nerves were still present in myenteric ganglia. Under nonadrenergic noncholinergic (NANC) conditions, small intestinal tissues from Cav1(-/-) mice relaxed to electrical field stimulation (EFS), as did tissues from control mice. Relaxation of tissues from control mice was markedly reduced by N-omega-nitro-L-arginine (10(-4) M), but relaxation of Cav1(-/-) animals was affected much less. Also, Cav1(-/-) mice tissues showed reduced relaxation responses to sodium nitroprusside (100 microM) compared to controls; yet there were no significant differences in the relaxation responses to 8-bromoguanosine-3': 5'-cyclic monophosphate (100 microM). Apamin (10(-6) M) significantly reduced relaxations to EFS in NANC conditions in Cav1(-/-) mice, but not in controls. The data from this study suggest that caveolin-1 gene knockout causes alterations in the smooth muscles and the ICC, leading to an impaired NO function in the mouse small intestine that could possibly be compensated by apamin-sensitive inhibitory mediators.
Figures

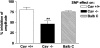
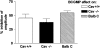
Similar articles
-
Impact of caveolin-1 knockout on NANC relaxation in circular muscles of the mouse small intestine compared with longitudinal muscles.Am J Physiol Gastrointest Liver Physiol. 2006 Feb;290(2):G394-403. doi: 10.1152/ajpgi.00321.2005. Epub 2005 Sep 15. Am J Physiol Gastrointest Liver Physiol. 2006. PMID: 16166342
-
Differential inhibitory control of circular and longitudinal smooth muscle layers of Balb/C mouse small intestine.Auton Neurosci. 2007 Jan 30;131(1-2):36-44. doi: 10.1016/j.autneu.2006.06.003. Epub 2006 Jul 17. Auton Neurosci. 2007. PMID: 16844426
-
A possible role of neurotensin in NANC relaxation of longitudinal muscle of the jejunum and ileum of Wistar rats.Br J Pharmacol. 2002 Nov;137(5):629-36. doi: 10.1038/sj.bjp.0704914. Br J Pharmacol. 2002. PMID: 12381676 Free PMC article.
-
Integrative Control of Gastrointestinal Motility by Nitric Oxide.Curr Med Chem. 2016;23(24):2715-2735. doi: 10.2174/0929867323666160812150907. Curr Med Chem. 2016. PMID: 27528058 Review.
-
Nitric Oxide: From Gastric Motility to Gastric Dysmotility.Int J Mol Sci. 2021 Sep 16;22(18):9990. doi: 10.3390/ijms22189990. Int J Mol Sci. 2021. PMID: 34576155 Free PMC article. Review.
Cited by
-
Human PTRF mutations cause secondary deficiency of caveolins resulting in muscular dystrophy with generalized lipodystrophy.J Clin Invest. 2009 Sep;119(9):2623-33. doi: 10.1172/JCI38660. Epub 2009 Aug 10. J Clin Invest. 2009. PMID: 19726876 Free PMC article.
-
Increased PDE5 activity and decreased Rho kinase and PKC activities in colonic muscle from caveolin-1-/- mice impair the peristaltic reflex and propulsion.Am J Physiol Gastrointest Liver Physiol. 2013 Dec;305(12):G964-74. doi: 10.1152/ajpgi.00165.2013. Epub 2013 Oct 24. Am J Physiol Gastrointest Liver Physiol. 2013. PMID: 24157969 Free PMC article.
-
NK receptors, Substance P, Ano1 expression and ultrastructural features of the muscle coat in Cav-1(-/-) mouse ileum.J Cell Mol Med. 2011 Nov;15(11):2411-20. doi: 10.1111/j.1582-4934.2011.01333.x. J Cell Mol Med. 2011. PMID: 21535398 Free PMC article.
-
Matrix metalloproteinase-2, caveolins, focal adhesion kinase and c-Kit in cells of the mouse myocardium.J Cell Mol Med. 2007 Sep-Oct;11(5):1069-86. doi: 10.1111/j.1582-4934.2007.00113.x. J Cell Mol Med. 2007. PMID: 17979883 Free PMC article.
-
Cavin-3 (PRKCDBP) deficiency reduces the density of caveolae in smooth muscle.Cell Tissue Res. 2017 Jun;368(3):591-602. doi: 10.1007/s00441-017-2587-y. Epub 2017 Mar 11. Cell Tissue Res. 2017. PMID: 28285351 Free PMC article.
References
-
- ANDRIES L., DUYMELINCK C., BORGERS M.Muscle and non-muscle markers in the interstitial cells of Cajal of guinea pig small intestine and colon Gastroenterology 2000118A4819(abstract)
-
- AZZENA G.B., MANCINELLI R. Nitric oxide regenerates the normal colonic peristaltic activity in mdx dystrophic mouse. Neurosci. Lett. 1999;261:9–12. - PubMed
-
- BAUER V., MATUSAK O. The non-adrenergic non-cholinergic innervation in the small intestine. Arch. Int. Pharmacodyn. Ther. 1986;280:137–163. - PubMed
-
- BRENMAN J.E., CHAO D.S., XIA H., ALDAPE K., BREDT D.S. Nitric oxide synthase complexed with dystrophin and absent from skeletal muscle sarcolemma in Duchenne muscle dystrophy. Cell. 1995;82:743–752. - PubMed
-
- BUCCI M., GRATTON J.-P., RUDIC R.D., ACEVEDO L., ROVIEZZO F., CIRINO G., SESSA W.C. In vivo delivery of caveolin-1 scaffolding domain inhibits nitric oxide synthesis and reduces inflammation. Nat. Med. 2000;6:1362–1367. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

