Randomised phase II evaluation of irinotecan plus high-dose 5-fluorouracil and leucovorin (ILF) vs 5-fluorouracil, leucovorin, and etoposide (ELF) in untreated metastatic gastric cancer
- PMID: 15942629
- PMCID: PMC2361806
- DOI: 10.1038/sj.bjc.6602649
Randomised phase II evaluation of irinotecan plus high-dose 5-fluorouracil and leucovorin (ILF) vs 5-fluorouracil, leucovorin, and etoposide (ELF) in untreated metastatic gastric cancer
Abstract
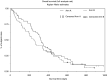
An open-label randomised comparison of efficacy and tolerability of irinotecan plus high-dose 5-fluorouracil (5-FU) and leucovorin (LV) (ILF) with etoposide plus 5-FU/LV (ELF) in patients with untreated metastatic or locally advanced gastric cancer. One cycle of ILF comprised six once-weekly infusions of irinotecan 80 mg m(-2), LV 500 mg m(-2), 24-h 5-FU 2000 mg m(-2), and ELF comprised three once-daily doses of etoposide 120 mg m(-2), LV 300 mg m(-2), 5-FU 500 mg m(-2). In all, 56 patients received ILF and 58 ELF. Median age was 62 years, Karnofsky performance 90%, and disease status was comparable for both arms. The objective clinical response rates after 14 weeks treatment (primary end point) were 30% for ILF and 17% for ELF (risk ratio (RR) 0.57, 95% confidence interval (CI) 0.29-1.13, P = 0.0766). Overall response rates over the entire treatment period for ILF and ELF were 43 and 24%, respectively (RR 0.56, 95% CI 0.33-0.97; P = 0.0467). For ILF and ELF, respectively, median progression-free survival was 4.5 vs 2.3 months, time to treatment failure was 3.6 vs 2.2 months (P = 0.4542), and overall survival was 10.8 vs 8.3 months (P = 0.2818). Both regimens were well tolerated, the main grade 3/4 toxicities being diarrhoea (18%, ILF) and neutropenia (57%, ELF). The data from this randomised phase II study indicate that ILF provides a better response rate than ELF, and that ILF should be investigated further for the treatment of metastatic gastric cancer.
Figures
References
-
- Assersohn L, Brown G, Cunningham D, Ward C, Oates J, Waters JS, Hill ME, Norman AR (2004) Phase II study of irinotecan and 5-fluorouracil/leucovorin in patients with primary refractory or relapsed advanced oesophageal and gastric carcinoma. Ann Oncol 15: 64–69 - PubMed
-
- Blanke CD, Haller DG, Benson AB, Rothenberg ML, Berlin J, Mori M, Hsieh YC, Miller LL (2001) A phase II study of irinotecan with 5-fluorouracil and leucovorin in patients with previously untreated gastric adenocarcinoma. Ann Oncol 12: 1575–1580 - PubMed
-
- Blot WJ, Devesa SS, Kneller RW, Fraumeni JFJ (1991) Rising incidence of adenocarcinoma of the esophagus and gastric cardia. JAMA 265: 1287–1289 - PubMed
-
- Bouche O, Raoul JL, Bonnetain F, Giovannini M, Etienne PL, Lledo G, Arsene D, Paitel JF, Guerin-Meyer V, Mitry E, Buecher B, Kaminsky MC, Seitz JF, Rougier P, Bedenne L, Milan C (2004) Randomized multicenter phase II trial of a biweekly regimen of fluorouracil and leucovorin (LV5FU2), LV5FU2 plus cisplatin, or LV5FU2 plus irinotecan in patients with previously untreated metastatic gastric cancer: a Federation Francophone de Cancerologie Digestive Group Study--FFCD 9803. J Clin Oncol 22: 4319–4328 - PubMed
-
- Bouzid K, Khalfallah S, Tujakowski J, Piko B, Purkalne G, Padrik P, Serafy M, Pshevloutsky EM, Boussard B (2003) A randomized phase II trial of irinotecan in combination with infusional or two different bolus 5-fluorouracil and folinic acid regimens as first-line therapy for advanced colorectal cancer. Ann Oncol 14: 1106–1114 - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical