Bone marrow-derived mesenchymal stem cells protect against experimental liver fibrosis in rats
- PMID: 15948250
- PMCID: PMC4315999
- DOI: 10.3748/wjg.v11.i22.3431
Bone marrow-derived mesenchymal stem cells protect against experimental liver fibrosis in rats
Abstract
Aim: Recent reports have shown the capacity of mesenchymal stem cells (MSCs) to differentiate into hepatocytes in vitro and in vivo. MSCs administration could repair injured liver, lung, or heart through reducing inflammation, collagen deposition, and remodeling. These results provide a clue to treatment of liver fibrosis. The aim of this study was to investigate the effect of infusion of bone marrow (BM)-derived MSCs on the experimental liver fibrosis in rats.
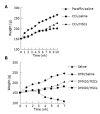
Methods: MSCs isolated from BM in male Fischer 344 rats were infused to female Wistar rats induced with carbon tetrachloride (CCl4) or dimethylnitrosamine (DMN). There were two random groups on the 42nd d of CCl4:CCl4/MSCs, to infuse a dose of MSCs alone; CCl4/saline, to infuse the same volume of saline as control. There were another three random groups after exposure to DMN: DMN10/MSCs, to infuse the same dose of MSCs on d 10; DMN10/saline, to infuse the same volume of saline on d 10; DMN20/MSCs, to infuse the same dose of MSCs on d 20. The morphological and behavioral changes of rats were monitored everyday. After 4-6 wk of MSCs administration, all rats were killed and fibrosis index were assessed by histopathology and radioimmunoassay. Smooth muscle alpha-actin (alpha-SMA) of liver were tested by immunohistochemistry and quantified by IBAS 2.5 software. Male rats sex determination region on the Y chromosome (sry) gene were explored by PCR.
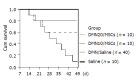
Results: Compared to controls, infusion of MSCs reduced the mortality rates of incidence in CCl4-induced model (10% vs 20%) and in DMN-induced model (20-40% vs 90%). The amount of collagen deposition and alpha-SMA staining was about 40-50% lower in liver of rats with MSCs than that of rats without MSCs. The similar results were observed in fibrosis index. And the effect of the inhibition of fibrogenesis was greater in DMN10/MSCs than in DMN20/MSCs. The sry gene was positive in the liver of rats with MSCs treatment by PCR.
Conclusion: MSCs treatment can protect against experimental liver fibrosis in CCl4-induced or DMN-induced rats and the mechanisms of the anti-fibrosis by MSCs will be studied further.
Figures







Similar articles
-
The therapeutic effects of bone marrow-derived mesenchymal stem cells and simvastatin in a rat model of liver fibrosis.Cell Biochem Biophys. 2014 Jan;68(1):111-25. doi: 10.1007/s12013-013-9698-1. Cell Biochem Biophys. 2014. PMID: 23807535
-
In vivo hepatic differentiation potential of human umbilical cord-derived mesenchymal stem cells: Therapeutic effect on liver fibrosis/cirrhosis.World J Gastroenterol. 2017 Dec 14;23(46):8152-8168. doi: 10.3748/wjg.v23.i46.8152. World J Gastroenterol. 2017. PMID: 29290652 Free PMC article.
-
Transplantation of human bone marrow mesenchymal stromal cells reduces liver fibrosis more effectively than Wharton's jelly mesenchymal stromal cells.Stem Cell Res Ther. 2017 Jun 13;8(1):143. doi: 10.1186/s13287-017-0595-1. Stem Cell Res Ther. 2017. PMID: 28610623 Free PMC article.
-
Transplantation of bone marrow-derived mesenchymal stem cells after regional hepatic irradiation ameliorates thioacetamide-induced liver fibrosis in rats.J Surg Res. 2014 Jan;186(1):408-16. doi: 10.1016/j.jss.2013.08.016. Epub 2013 Sep 11. J Surg Res. 2014. PMID: 24071025
-
[Research progress of endogenous bone marrow-derived mesenchymal stem cells in pulmonary fibrosis].Sheng Li Xue Bao. 2020 Oct 25;72(5):597-604. Sheng Li Xue Bao. 2020. PMID: 33106830 Review. Chinese.
Cited by
-
Effectiveness and mechanisms of mesenchymal stem cell therapy in preclinical animal models of hepatic fibrosis: a systematic review and meta-analysis.Front Bioeng Biotechnol. 2024 Jul 22;12:1424253. doi: 10.3389/fbioe.2024.1424253. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39104627 Free PMC article.
-
Embryonic stem cells develop into hepatocytes after intrasplenic transplantation in CCl4-treated mice.World J Gastroenterol. 2007 Feb 14;13(6):866-73. doi: 10.3748/wjg.v13.i6.866. World J Gastroenterol. 2007. PMID: 17352015 Free PMC article.
-
Cellular therapy for liver disease.Mayo Clin Proc. 2014 Mar;89(3):414-24. doi: 10.1016/j.mayocp.2013.10.023. Mayo Clin Proc. 2014. PMID: 24582199 Free PMC article. Review.
-
Stem Cell Therapy: Current Applications and Potential for Urology.Curr Urol Rep. 2015 Nov;16(11):77. doi: 10.1007/s11934-015-0551-5. Curr Urol Rep. 2015. PMID: 26385812 Review.
-
Stem cell therapy in bladder dysfunction: where are we? And where do we have to go?Biomed Res Int. 2013;2013:930713. doi: 10.1155/2013/930713. Epub 2013 Sep 16. Biomed Res Int. 2013. PMID: 24151627 Free PMC article. Review.
References
-
- Friedman SL. Liver fibrosis -- from bench to bedside. J Hepatol. 2003;38 Suppl 1:S38–S53. - PubMed
-
- Lee DS, Gil WH, Lee HH, Lee KW, Lee SK, Kim SJ, Choi SH, Heo JS, Hyon WS, Kim GS, et al. Factors affecting graft survival after living donor liver transplantation. Transplant Proc. 2004;36:2255–2256. - PubMed
-
- Wen GM, Li HW, Xiao QZ, Cheng ZG, Zhang XM, Li Y, Duan LN, Li SN. Studies on differentiation potential of human bone marrow mesenchymal stem cells into hematopoietic cells in vivo. Zhongguo Bingli Shengli Zazhi. 2003;19:157–162.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

