Characterization of vestibular dysfunction in the mouse model for Usher syndrome 1F
- PMID: 15952048
- PMCID: PMC2538330
- DOI: 10.1007/s10162-004-5032-3
Characterization of vestibular dysfunction in the mouse model for Usher syndrome 1F
Abstract
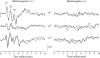
The deaf-circling Ames waltzer (av) mouse harbors a mutation in the protocadherin 15 (Pcdh15) gene and is a model for inner ear defects associated with Usher syndrome type 1F. Earlier studies showed altered cochlear hair cell morphology in young av mice. In contrast, no structural abnormality consistent with significant vestibular dysfunction in young av mice was observed. Light and scanning electron microscopic studies showed that vestibular hair cells from presumptive null alleles Pcdh15(av-Tg) and Pcdh15(av-3J) are morphologically similar to vestibular sensory cells from control littermates, suggesting that the observed phenotype in these alleles might be a result of a central, rather than peripheral, defect. In the present study, a combination of physiologic and anatomic methods was used to more thoroughly investigate the source of vestibular dysfunction in Ames waltzer mice. Analysis of vestibular evoked potentials and angular vestibulo-ocular reflexes revealed a lack of physiologic response to linear and angular acceleratory stimuli in Pcdh15 mutant mice. Optokinetic reflex function was diminished but still present in the mutant animals, suggesting that the defect is primarily peripheral in nature. These findings indicate that the mutation in Pcdh15 results in either a functional abnormality in the vestibular receptor organs or that the defects are limited to the vestibular nerve. AM1-43 dye uptake has been shown to correlate with normal transduction function in hair cells. Dye uptake was found to be dramatically reduced in Pcdh15 mutants compared to control littermates, suggesting that the mutation affects hair cell function, although structural abnormalities consistent with significant vestibular dysfunction are not apparent by light and scanning electron microscopy in the vestibular neuroepithelia of young animals.
Figures





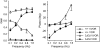


References
-
- Ahmed ZM, Riazuddin S, Ahmad J, Bernstein SL, Guo Y, Sabar MF, Sieving P, Griffith AJ, Friedman TB, Belyantseva IA, Wilcox ER. PCDH15 is expressed in the neurosensory epithelium of the eye and ear and mutant alleles are responsible for both USH1F and DFNB23. Hum. Mol. Genet. 2003;12:3215–3223. - PubMed
-
- Alagramam KN, Zahorsky-Reeves J, Wright CG, Pawlowski KS, Erway LC, Stubbs L, Woychik RP. Neuroepithelial defects of the inner ear in a new allele of the mouse mutation Ames waltzer. Hear. Res. 2000;148:181–191. - PubMed
-
- Alagramam KN, Murcia CL, Kwon HY, Pawlowski KS, Wright CG, Woychik RP. The mouse Ames waltzer hearing-loss mutant is caused by mutation of PCDH15, a novel protocadherin gene. Nat. Genet. 2001;27:99–102. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

