Participation of both Gab1 and Gab2 in the activation of the ERK/MAPK pathway by epidermal growth factor
- PMID: 15952937
- PMCID: PMC1237148
- DOI: 10.1042/BJ20050229
Participation of both Gab1 and Gab2 in the activation of the ERK/MAPK pathway by epidermal growth factor
Abstract
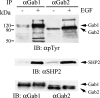
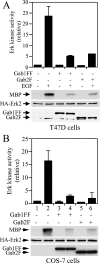
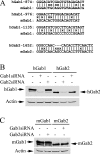
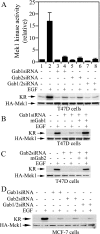
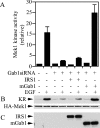
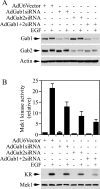
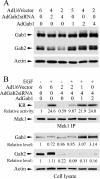
Three members of Gab family docking proteins, Gab1, Gab2 and Gab3, have been identified in humans. Previous studies have found that the hepatocyte growth factor preferentially utilizes Gab1 for signalling, whereas Bcr-Abl selectively signals through Gab2. Gab1-SHP2 interaction has been shown to mediate ERK (extracellular-signal-regulated kinase) activation by EGF (epidermal growth factor). However, it was unclear whether EGF selectively utilizes Gab1 for signalling to ERK and whether Gab2 is dispensable in cells where Gab1 and Gab2 are co-expressed. Using T47D and MCF-7 human breast carcinoma cells that express endogenous Gab1 and Gab2, we examined the role of these docking proteins in EGF-induced ERK activation. It was found that EGF induced a similar amount of SHP2-Gab1 and SHP2-Gab2 complexes. Expression of either SHP2-binding defective Gab1 or Gab2 mutant blocked EGF-induced ERK activation. Down-regulation of either Gab1 or Gab2 by siRNAs (small interfering RNAs) effectively inhibited the EGF-stimulated ERK activation pathway and cell migration. Interestingly, the inhibitory effect of Gab1 siRNA could be rescued not only by expression of an exogenous mouse Gab1 but also by an exogenous human Gab2 and vice versa, but not by IRS1 (insulin receptor substrate 1). These results reveal that Gab2 plays a pivotal role in the EGF-induced ERK activation pathway and that it can complement the function of Gab1 in the EGF signalling pathway. Furthermore, Gab1 and Gab2 are critical signalling threshold proteins for ERK activation by EGF.
Figures


Similar articles
-
Interleukin 6 inhibits proliferation and, in cooperation with an epidermal growth factor receptor autocrine loop, increases migration of T47D breast cancer cells.Cancer Res. 2001 Jan 1;61(1):383-91. Cancer Res. 2001. PMID: 11196191
-
Roles of Gab1 and SHP2 in paxillin tyrosine dephosphorylation and Src activation in response to epidermal growth factor.J Biol Chem. 2004 Feb 27;279(9):8497-505. doi: 10.1074/jbc.M312575200. Epub 2003 Dec 8. J Biol Chem. 2004. PMID: 14665621
-
The involvement of the docking protein Gab1 in mitogenic signalling induced by EGF and HGF in rat hepatocytes.Biochim Biophys Acta. 2013 Dec;1833(12):3286-3294. doi: 10.1016/j.bbamcr.2013.10.004. Epub 2013 Oct 12. Biochim Biophys Acta. 2013. PMID: 24126105
-
Gab-family adapter molecules in signal transduction of cytokine and growth factor receptors, and T and B cell antigen receptors.Leuk Lymphoma. 2000 Apr;37(3-4):299-307. doi: 10.3109/10428190009089430. Leuk Lymphoma. 2000. PMID: 10752981 Review.
-
Essential roles of Gab1 tyrosine phosphorylation in growth factor-mediated signaling and angiogenesis.Int J Cardiol. 2015 Feb 15;181:180-4. doi: 10.1016/j.ijcard.2014.10.148. Epub 2014 Oct 24. Int J Cardiol. 2015. PMID: 25528308 Free PMC article. Review.
Cited by
-
FoxO1-GAB1 axis regulates homing capacity and tonic AKT activity in chronic lymphocytic leukemia.Blood. 2021 Sep 2;138(9):758-772. doi: 10.1182/blood.2020008101. Blood. 2021. PMID: 33786575 Free PMC article.
-
Function, regulation and pathological roles of the Gab/DOS docking proteins.Cell Commun Signal. 2009 Sep 8;7:22. doi: 10.1186/1478-811X-7-22. Cell Commun Signal. 2009. PMID: 19737390 Free PMC article.
-
Effect of a neurokinin-1 receptor antagonist in a rat model of colitis-associated colon cancer.Anticancer Res. 2010 Sep;30(9):3345-53. Anticancer Res. 2010. PMID: 20944107 Free PMC article.
-
The Gab2 in signal transduction and its potential role in the pathogenesis of Alzheimer's disease.Neurosci Bull. 2010 Jun;26(3):241-6. doi: 10.1007/s12264-010-1109-7. Neurosci Bull. 2010. PMID: 20502503 Free PMC article. Review.
-
Systems-level interactions between insulin-EGF networks amplify mitogenic signaling.Mol Syst Biol. 2009;5:256. doi: 10.1038/msb.2009.19. Epub 2009 Apr 7. Mol Syst Biol. 2009. PMID: 19357636 Free PMC article.
References
-
- Neel B. G., Gu H., Pao L. The ‘Shp’ing news: SH2 domain-containing tyrosine phosphatases in cell signaling. Trends Biochem. Sci. 2003;28:284–293. - PubMed
-
- Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell (Cambridge, Mass.) 2000;103:211–225. - PubMed
-
- Feng G. S. Shp-2 tyrosine phosphatase: signaling one cell or many. Exp. Cell Res. 1999;253:47–54. - PubMed
-
- Cunnick J. M., Dorsey J. F., Munoz-Antonia T., Mei L., Wu J. Requirement of SHP2 binding to Grb2-associated binder-1 for mitogen-activated protein kinase activation in response to lysophosphatidic acid and epidermal growth factor. J. Biol. Chem. 2000;275:13842–13848. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

