Identification and characterization of molecular interactions between mortalin/mtHsp70 and HSP60
- PMID: 15957980
- PMCID: PMC1276915
- DOI: 10.1042/BJ20050861
Identification and characterization of molecular interactions between mortalin/mtHsp70 and HSP60
Abstract
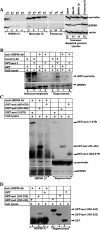
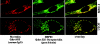
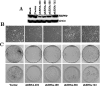
Mortalin/mtHsp70 (mitochondrial Hsp70) and HSP60 (heat-shock protein 60) are heat-shock proteins that reside in multiple subcellular compartments, with mitochondria being the predominant one. In the present study, we demonstrate that the two proteins interact both in vivo and in vitro, and that the N-terminal region of mortalin is involved in these interactions. Suppression of HSP60 expression by shRNA (short hairpin RNA) plasmids caused the growth arrest of cancer cells similar to that obtained by suppression of mortalin expression by ribozymes. An overexpression of mortalin, but not of HSP60, extended the in vitro lifespan of normal fibroblasts (TIG-1). Taken together, this study for the first time delineates: (i) molecular interactions of HSP60 with mortalin; (ii) their co- and exclusive localizations in vivo; (iii) their involvement in tumorigenesis; and (iv) their functional distinction in pathways involved in senescence.
Figures

Similar articles
-
Quantum dot-based protein imaging and functional significance of two mitochondrial chaperones in cellular senescence and carcinogenesis.Ann N Y Acad Sci. 2006 May;1067:469-73. doi: 10.1196/annals.1354.067. Ann N Y Acad Sci. 2006. PMID: 16804028
-
On the brotherhood of the mitochondrial chaperones mortalin and heat shock protein 60.Cell Stress Chaperones. 2006 Summer;11(2):116-28. doi: 10.1379/csc-144r.1. Cell Stress Chaperones. 2006. PMID: 16817317 Free PMC article. Review.
-
Reduction in mortalin level by its antisense expression causes senescence-like growth arrest in human immortalized cells.J Gene Med. 2004 Apr;6(4):439-44. doi: 10.1002/jgm.530. J Gene Med. 2004. PMID: 15079818
-
Mitochondrial heat shock protein (HSP) 70 synergizes with HSP60 in transducing endothelial cell apoptosis induced by anti-HSP60 autoantibody.FASEB J. 2009 Aug;23(8):2772-9. doi: 10.1096/fj.08-128785. Epub 2009 Apr 3. FASEB J. 2009. PMID: 19346294
-
Involvement of mortalin in cellular senescence from the perspective of its mitochondrial import, chaperone, and oxidative stress management functions.Ann N Y Acad Sci. 2007 Apr;1100:306-11. doi: 10.1196/annals.1395.032. Ann N Y Acad Sci. 2007. PMID: 17460192 Review.
Cited by
-
CCAR2/DBC1 and Hsp60 Positively Regulate Expression of Survivin in Neuroblastoma Cells.Int J Mol Sci. 2019 Jan 1;20(1):131. doi: 10.3390/ijms20010131. Int J Mol Sci. 2019. PMID: 30609639 Free PMC article.
-
Chaperones and Beyond as Key Players in Pluripotency Maintenance.Front Cell Dev Biol. 2019 Aug 2;7:150. doi: 10.3389/fcell.2019.00150. eCollection 2019. Front Cell Dev Biol. 2019. PMID: 31428613 Free PMC article. Review.
-
Chaperones vs. oxidative stress in the pathobiology of ischemic stroke.Front Mol Neurosci. 2024 Dec 11;17:1513084. doi: 10.3389/fnmol.2024.1513084. eCollection 2024. Front Mol Neurosci. 2024. PMID: 39723236 Free PMC article. Review.
-
Heat shock protein paradigms in cancer progression: future therapeutic perspectives.3 Biotech. 2024 Apr;14(4):96. doi: 10.1007/s13205-024-03951-6. Epub 2024 Mar 5. 3 Biotech. 2024. PMID: 38449709 Free PMC article. Review.
-
Protein homeostasis and aging: The importance of exquisite quality control.Ageing Res Rev. 2011 Apr;10(2):205-15. doi: 10.1016/j.arr.2010.02.001. Epub 2010 Feb 10. Ageing Res Rev. 2011. PMID: 20152936 Free PMC article. Review.
References
-
- Webster T. J., Naylor D. J., Hartman D. J., Hoj P. B., Hoogenraad N. J. cDNA cloning and efficient mitochondrial import of pre-mtHSP70 from rat liver. DNA Cell Biol. 1994;13:1213–1220. - PubMed
-
- Michikawa Y., Baba T., Arai Y., Sakakura T., Kusakabe M. Structure and organization of the gene encoding a mouse mitochondrial stress-70 protein. FEBS Lett. 1993;336:27–33. - PubMed
-
- Wadhwa R., Kaul S. C., Mitsui Y., Sugimoto Y. Differential subcellular distribution of mortalin in mortal and immortal mouse and human fibroblasts. Exp. Cell Res. 1993;207:442–448. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

