Gastrointestinal mucins of Fut2-null mice lack terminal fucosylation without affecting colonization by Candida albicans
- PMID: 15958416
- PMCID: PMC1360228
- DOI: 10.1093/glycob/cwi089
Gastrointestinal mucins of Fut2-null mice lack terminal fucosylation without affecting colonization by Candida albicans
Abstract
Posttranslational modification of apomucins by the sequential action of glycosyltransferases is required to produce mature mucins. The Secretor gene (FUT2) encodes an alpha(1,2)fucosyltransferase (EC 2.4.1.69) that catalyzes addition of terminal alpha(1,2)fucose residues on mucins and other molecules in mucosal epithelium. Mutant mice containing targeted replacement of Fut2 with the bacterial reporter gene lacZ were studied to determine the affect of the loss of Fut2 on glycosylation of mucins in the gastrointestinal tract. By whole organ X-gal staining, lacZ activity is prominently expressed in the foveolar pit and chief cells of the glandular stomach, Brunner's glands of the duodenum, and goblet cells in the large intestine of Fut2-LacZ-null mice. Staining with Aleuria aurantia agglutinin demonstrates loss of L-fucosylated epithelial glycans throughout the gastrointestinal tract of Fut2-LacZ-null mice, however, histologic appearance of the tissues appears normal. Analysis of oligosaccharides released from insoluble colonic mucins, largely Muc2, by mass spectrometry shows complete lack of terminal fucosylation of O-linked oligosaccharides in Fut2-LacZ-null mice. Precursor glycans accumulate with no evidence of compensation by other fucosyltransferases or sialyltransferases on mucin glycosylation. Because Candida albicans has been reported to adhere to intestinal mucins creating a potential reservoir associated with vaginitis, Fut2-LacZ-null and wild-type mice were inoculated by gastric lavage with C. albicans. We observe no difference in colonization between genotypes suggesting mucin terminal fucosylation does not significantly influence C. albicans-host interaction in the intestine, highlighting that infections caused by the same organism at different mucosal surfaces are not equal.
Figures
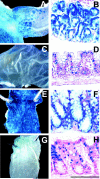
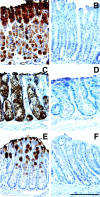

Similar articles
-
Mucin-type O-glycans and their roles in intestinal homeostasis.Glycobiology. 2013 Sep;23(9):1026-37. doi: 10.1093/glycob/cwt045. Epub 2013 Jun 10. Glycobiology. 2013. PMID: 23752712 Free PMC article. Review.
-
Cervical mucins carry alpha(1,2)fucosylated glycans that partly protect from experimental vaginal candidiasis.Glycoconj J. 2009 Dec;26(9):1125-34. doi: 10.1007/s10719-009-9234-0. Glycoconj J. 2009. PMID: 19326211 Free PMC article.
-
LacZ expression in Fut2-LacZ reporter mice reveals estrogen-regulated endocervical glandular expression during estrous cycle, hormone replacement, and pregnancy.Glycobiology. 2004 Feb;14(2):169-75. doi: 10.1093/glycob/cwh019. Epub 2003 Oct 23. Glycobiology. 2004. PMID: 14576173 Free PMC article.
-
Tissue-specific loss of fucosylated glycolipids in mice with targeted deletion of alpha(1,2)fucosyltransferase genes.Biochem J. 2004 May 15;380(Pt 1):75-81. doi: 10.1042/BJ20031668. Biochem J. 2004. PMID: 14967068 Free PMC article.
-
Brunner's glands: a structural, histochemical and pathological profile.Prog Histochem Cytochem. 2000;35(4):259-367. Prog Histochem Cytochem. 2000. PMID: 11148980 Review.
Cited by
-
Mucin-type O-glycans and their roles in intestinal homeostasis.Glycobiology. 2013 Sep;23(9):1026-37. doi: 10.1093/glycob/cwt045. Epub 2013 Jun 10. Glycobiology. 2013. PMID: 23752712 Free PMC article. Review.
-
Genome-wide association meta-analysis yields 20 loci associated with gallstone disease.Nat Commun. 2018 Nov 30;9(1):5101. doi: 10.1038/s41467-018-07460-y. Nat Commun. 2018. PMID: 30504769 Free PMC article.
-
Enterocytes, fibroblasts and myeloid cells synergize in anti-bacterial and anti-viral pathways with IL22 as the central cytokine.Commun Biol. 2021 May 27;4(1):631. doi: 10.1038/s42003-021-02176-0. Commun Biol. 2021. PMID: 34045640 Free PMC article.
-
Intestinal Goblet Cell Loss during Chorioamnionitis in Fetal Lambs: Mechanistic Insights and Postnatal Implications.Int J Mol Sci. 2021 Feb 16;22(4):1946. doi: 10.3390/ijms22041946. Int J Mol Sci. 2021. PMID: 33669331 Free PMC article.
-
Antenatal Ureaplasma infection induces ovine small intestinal goblet cell defects: a strong link with NEC pathology.Tissue Barriers. 2023 Oct 2;11(4):2158016. doi: 10.1080/21688370.2022.2158016. Epub 2022 Dec 28. Tissue Barriers. 2023. PMID: 36576242 Free PMC article.
References
-
- Bry L, Falk PG, Midtvedt T, Gordon JI. A model of host-microbial interactions in an open mammalian ecosystem. Science. 1996;273:1380–1383. - PubMed
-
- Carlstedt I, Herrmann A, Karlsson H, Sheehan J, Fransson LA, Hansson GC. Characterization of two different glycosylated domains from the insoluble mucin complex of rat small intestine. J Biol Chem. 1993;268:18771–18781. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

