Nucleotide-dependent bending flexibility of tubulin regulates microtubule assembly
- PMID: 15959508
- PMCID: PMC1386036
- DOI: 10.1038/nature03606
Nucleotide-dependent bending flexibility of tubulin regulates microtubule assembly
Abstract
The atomic structure of tubulin in a polymerized, straight protofilament is clearly distinct from that in a curved conformation bound to a cellular depolymerizer. The nucleotide contents are identical, and in both cases the conformation of the GTP-containing, intra-dimer interface is indistinguishable from the GDP-containing, inter-dimer contact. Here we present two structures corresponding to the start and end points in the microtubule polymerization and hydrolysis cycles that illustrate the consequences of nucleotide state on longitudinal and lateral assembly. In the absence of depolymerizers, GDP-bound tubulin shows distinctive intra-dimer and inter-dimer interactions and thus distinguishes the GTP and GDP interfaces. A cold-stable tubulin polymer with the non-hydrolysable GTP analogue GMPCPP, containing semi-conserved lateral interactions, supports a model in which the straightening of longitudinal interfaces happens sequentially, starting with a conformational change after GTP binding that straightens the dimer enough for the formation of lateral contacts into a non-tubular intermediate. Closure into a microtubule does not require GTP hydrolysis.
Conflict of interest statement
The authors have no financial interest concerning this work.
Figures


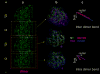
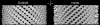
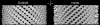
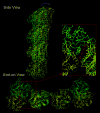


Comment in
-
Cell biology: powerful curves.Nature. 2005 Jun 16;435(7044):895-7. doi: 10.1038/435895a. Nature. 2005. PMID: 15959501 No abstract available.
References
-
- Desai A, Mitchison TJ. Microtubule polymerization dynamics. Annu Rev Cell Dev Biol. 1997;13:83–117. - PubMed
-
- Jordan MA. Mechanism of action of antitumor drugs that interact with microtubules and tubulin. Curr Med Chem Anti-Canc Agents. 2002;2:1–17. - PubMed
-
- Heald R, Nogales E. Microtubule dynamics. J Cell Sci. 2002;115:3–4. - PubMed
-
- Nogales E, Wolf SG, Downing KH. Structure of the αβ tubulin dimer by electron crystallography. Nature. 1998;391:199–203. - PubMed
-
- Löwe J, Li H, Downing KH, Nogales E. Refined structure of αβ-tubulin at 3.5 Å resolution. J Mol Biol. 2001;313:1045–1057. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

