Differential effects of proteasome inhibition by bortezomib on murine acute graft-versus-host disease (GVHD): delayed administration of bortezomib results in increased GVHD-dependent gastrointestinal toxicity
- PMID: 15961519
- PMCID: PMC1895334
- DOI: 10.1182/blood-2004-11-4526
Differential effects of proteasome inhibition by bortezomib on murine acute graft-versus-host disease (GVHD): delayed administration of bortezomib results in increased GVHD-dependent gastrointestinal toxicity
Abstract
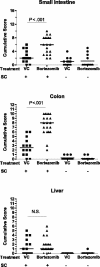
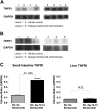
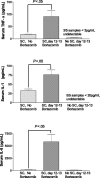
We have recently demonstrated that the proteasome inhibitor, bortezomib, administered immediately following murine allogeneic bone marrow transplantation (BMT) resulted in marked inhibition of acute graft-versus-host disease (GVHD) with retention of graft-versus-tumor effects. We now assessed the effects of delayed bortezomib administration (5 or more days after BMT) on GVHD. Recipient C57BL/6 (H2b) mice were lethally irradiated and given transplants of bone marrow cells and splenocytes from major histocompatibility complex (MHC)-disparate BALB/c (H2d) donors. In marked contrast to the effects of bortezomib on GVHD prevention when administered immediately after BMT, delayed bortezomib administration resulted in significant acceleration of GVHD-dependent morbidity. No toxicity was observed following delayed bortezomib administration in models where donor T cells were not coadministered, indicating that these deleterious effects were critically dependent on GVHD induction. The increase in GVHD susceptibility even occurred when late administration of bortezomib was preceded by early administration. Pathologic assessment revealed that significant increases in gastrointestinal lesions occurred following delayed bortezomib administration during GVHD. This pathology correlated with significant increases of type 1 tumor necrosis factor alpha (TNF-alpha) receptor transcription in gastrointestinal cells and with significant increases of TNF-alpha, interleukin 1beta (IL-1beta), and IL-6 levels in the serum. These results indicate that the differential effects of proteasome inhibition with bortezomib on GVHD are critically dependent on the timing of bortezomib administration.
Figures



Similar articles
-
IL-1β and TLR4 signaling are involved in the aggravated murine acute graft-versus-host disease caused by delayed bortezomib administration.J Immunol. 2014 Feb 1;192(3):1277-85. doi: 10.4049/jimmunol.1203428. Epub 2013 Dec 20. J Immunol. 2014. PMID: 24363427
-
Inhibition of acute graft-versus-host disease with retention of graft-versus-tumor effects by the proteasome inhibitor bortezomib.Proc Natl Acad Sci U S A. 2004 May 25;101(21):8120-5. doi: 10.1073/pnas.0401563101. Epub 2004 May 17. Proc Natl Acad Sci U S A. 2004. PMID: 15148407 Free PMC article.
-
Differential effects of donor T-cell cytokines on outcome with continuous bortezomib administration after allogeneic bone marrow transplantation.Blood. 2008 Aug 15;112(4):1522-9. doi: 10.1182/blood-2008-03-143461. Epub 2008 Jun 6. Blood. 2008. PMID: 18539902 Free PMC article.
-
Proteasome inhibition in transplantation-focusing on the experience with bortezomib.Curr Pharm Des. 2013;19(18):3299-304. doi: 10.2174/13816128113199990308. Curr Pharm Des. 2013. PMID: 23151133 Review.
-
Proteasome inhibitors.Prog Mol Biol Transl Sci. 2012;109:161-226. doi: 10.1016/B978-0-12-397863-9.00005-5. Prog Mol Biol Transl Sci. 2012. PMID: 22727422 Review.
Cited by
-
Prospective phase 2 trial of ixazomib after nonmyeloablative haploidentical peripheral blood stem cell transplant.Blood Adv. 2020 Aug 11;4(15):3669-3676. doi: 10.1182/bloodadvances.2020001958. Blood Adv. 2020. PMID: 32777064 Free PMC article. Clinical Trial.
-
Graft-Versus-Host Disease Mouse Models: A Clinical-Translational Perspective.Methods Mol Biol. 2025;2907:1-56. doi: 10.1007/978-1-0716-4430-0_1. Methods Mol Biol. 2025. PMID: 40100591 Review.
-
Bortezomib-based graft-versus-host disease prophylaxis in HLA-mismatched unrelated donor transplantation.J Clin Oncol. 2012 Sep 10;30(26):3202-8. doi: 10.1200/JCO.2012.42.0984. Epub 2012 Aug 6. J Clin Oncol. 2012. PMID: 22869883 Free PMC article. Clinical Trial.
-
Emerging therapies in hematopoietic stem cell transplantation.Biol Blood Marrow Transplant. 2012 Jan;18(1 Suppl):S125-31. doi: 10.1016/j.bbmt.2011.10.022. Biol Blood Marrow Transplant. 2012. PMID: 22226096 Free PMC article. Review.
-
Role of allogeneic transplantation in multiple myeloma in the era of new drugs.Mediterr J Hematol Infect Dis. 2010 Jun 1;2(2):e2010013. doi: 10.4084/MJHID.2010.013. Mediterr J Hematol Infect Dis. 2010. PMID: 21415966 Free PMC article.
References
-
- Ferrara JL, Deeg H, Burakoff SJ. Graft-versus-Host Disease. Marcel Decker: New York, NY; 1997.
-
- Ferrara JL. Pathogenesis of acute graft-versus-host disease: cytokines and cellular effectors. J Hematother Stem Cell Res. 2000;9: 299-306. - PubMed
-
- Ferrara JL, Cooke KR, Teshima T. The pathophysiology of acute graft-versus-host disease. Int J Hematol. 2003;78: 181-187. - PubMed
-
- Goldberg AL, Rock K. Not just research tools— proteasome inhibitors offer therapeutic promise. Nat Med. 2002;8: 338-340. - PubMed
-
- Adams J, Kauffman M. Development of the proteasome inhibitor Velcade (bortezomib). Cancer Invest. 2004;22: 304-311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

