Intravenous apoptotic spleen cell infusion induces a TGF-beta-dependent regulatory T-cell expansion
- PMID: 15962005
- PMCID: PMC3448559
- DOI: 10.1038/sj.cdd.4401699
Intravenous apoptotic spleen cell infusion induces a TGF-beta-dependent regulatory T-cell expansion
Abstract
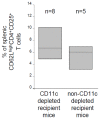
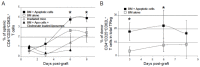
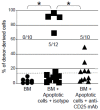
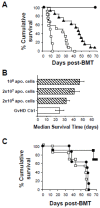
Apoptotic leukocytes are endowed with immunomodulatory properties that can be used to enhance hematopoietic engraftment and prevent graft-versus-host disease (GvHD). This apoptotic cell-induced tolerogenic effect is mediated by host macrophages and not recipient dendritic cells or donor phagocytes present in the bone marrow graft as evidenced by selective cell depletion and trafficking experiments. Furthermore, apoptotic cell infusion is associated with TGF-beta-dependent donor CD4+CD25+ T-cell expansion. Such cells have a regulatory phenotype (CD62L(high) and intracellular CTLA-4+), express high levels of forkhead-box transcription factor p3 (Foxp3) mRNA and exert ex vivo suppressive activity through a cell-to-cell contact mechanism. In vivo CD25 depletion after apoptotic cell infusion prevents the apoptotic cell-induced beneficial effects on engraftment and GvHD occurrence. This highlights the role of regulatory T cells in the tolerogenic effect of apoptotic cell infusion. This novel association between apoptosis and regulatory T-cell expansion may also contribute to preventing deleterious autoimmune responses during normal turnover.
Figures



Similar articles
-
IL-2-targeted therapy ameliorates the severity of graft-versus-host disease: ex vivo selective depletion of host-reactive T cells and in vivo therapy.Biol Blood Marrow Transplant. 2012 Apr;18(4):523-35. doi: 10.1016/j.bbmt.2011.11.016. Epub 2012 Jan 3. Biol Blood Marrow Transplant. 2012. PMID: 22227590
-
Plasmacytoid dendritic cells play a major role in apoptotic leukocyte-induced immune modulation.J Immunol. 2011 May 15;186(10):5696-705. doi: 10.4049/jimmunol.1001523. Epub 2011 Apr 1. J Immunol. 2011. PMID: 21460208
-
Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3.J Exp Med. 2003 Dec 15;198(12):1875-86. doi: 10.1084/jem.20030152. J Exp Med. 2003. PMID: 14676299 Free PMC article.
-
Role of naturally arising regulatory T cells in hematopoietic cell transplantation.Biol Blood Marrow Transplant. 2006 Oct;12(10):995-1009. doi: 10.1016/j.bbmt.2006.04.009. Biol Blood Marrow Transplant. 2006. PMID: 17084366 Review.
-
Dendritic cells expand antigen-specific Foxp3+ CD25+ CD4+ regulatory T cells including suppressors of alloreactivity.Immunol Rev. 2006 Aug;212:314-29. doi: 10.1111/j.0105-2896.2006.00422.x. Immunol Rev. 2006. PMID: 16903923 Review.
Cited by
-
Regulatory T-cell immunotherapy for allogeneic hematopoietic stem-cell transplantation.Ther Adv Hematol. 2012 Feb;3(1):29-44. doi: 10.1177/2040620711422266. Ther Adv Hematol. 2012. PMID: 23556110 Free PMC article.
-
Peripheral blood mononuclear cell secretome for tissue repair.Apoptosis. 2016 Dec;21(12):1336-1353. doi: 10.1007/s10495-016-1292-8. Apoptosis. 2016. PMID: 27696124 Free PMC article. Review.
-
Determining the effector response to cell death.Nat Rev Immunol. 2021 May;21(5):292-304. doi: 10.1038/s41577-020-00456-0. Epub 2020 Nov 13. Nat Rev Immunol. 2021. PMID: 33188303 Free PMC article. Review.
-
Apoptotic cell-linked immunoregulation: implications for promoting immune tolerance in transplantation.Cell Biosci. 2015 Jun 7;5:27. doi: 10.1186/s13578-015-0019-9. eCollection 2015. Cell Biosci. 2015. PMID: 26110047 Free PMC article.
-
Selective depletion of CD11c+ CD11b+ dendritic cells partially abrogates tolerogenic effects of intravenous MOG in murine EAE.Eur J Immunol. 2016 Oct;46(10):2454-2466. doi: 10.1002/eji.201546274. Eur J Immunol. 2016. PMID: 27338697 Free PMC article.
References
-
- Kamradt T, Mitchison NA. Tolerance and autoimmunity. N Engl J Med. 2001;344:655–64. - PubMed
-
- Sakaguchi S. Regulatory T cells: key controllers of immunologic self-tolerance. Cell. 2000;101:455–8. - PubMed
-
- Shevach EM. CD4+ CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol. 2002;2:389–400. - PubMed
-
- Wood KJ, Sakaguchi S. Regulatory T cells in transplantation tolerance. Nat Rev Immunol. 2003;3:199–210. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

