The virion N protein of infectious bronchitis virus is more phosphorylated than the N protein from infected cell lysates
- PMID: 15979680
- PMCID: PMC7111880
- DOI: 10.1016/j.virol.2005.04.029
The virion N protein of infectious bronchitis virus is more phosphorylated than the N protein from infected cell lysates
Abstract
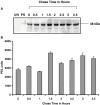
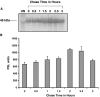
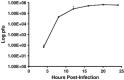
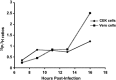
Because phosphorylation of the infectious bronchitis virus (IBV) nucleocapsid protein (N) may regulate its multiple roles in viral replication, the dynamics of N phosphorylation were examined. 32P-orthophosphate labeling and Western blot analyses confirmed that N was the only viral protein that was phosphorylated. Pulse labeling with 32P-orthophosphate indicated that the IBV N protein was phosphorylated in the virion, as well as at all times during infection in either chicken embryo kidney cells or Vero cells. Pulse-chase analyses followed by immunoprecipitation of IBV N proteins using rabbit anti-IBV N polyclonal antibody demonstrated that the phosphate on the N protein was stable for at least 1 h. Simultaneous labeling with 32P-orthophosphate and 3H-leucine identified a 3.5-fold increase in the 32P:3H counts per minute (cpm) ratio of N in the virion as compared to the 32P:3H cpm ratio of N in the cell lysates from chicken embryo kidney cells, whereas in Vero cells the 32P:3H cpm ratio of N from the virion was 10.5-fold greater than the 32P:3H cpm ratio of N from the cell lysates. These studies are consistent with the phosphorylation of the IBV N playing a role in assembly or maturation of the viral particle.
Figures


Similar articles
-
Identification of two ATR-dependent phosphorylation sites on coronavirus nucleocapsid protein with nonessential functions in viral replication and infectivity in cultured cells.Virology. 2013 Sep;444(1-2):225-32. doi: 10.1016/j.virol.2013.06.014. Epub 2013 Jul 9. Virology. 2013. PMID: 23849791 Free PMC article.
-
Role of phosphorylation clusters in the biology of the coronavirus infectious bronchitis virus nucleocapsid protein.Virology. 2008 Jan 20;370(2):373-81. doi: 10.1016/j.virol.2007.08.016. Epub 2007 Nov 1. Virology. 2008. PMID: 17931676 Free PMC article.
-
Characterization of cellular furin content as a potential factor determining the susceptibility of cultured human and animal cells to coronavirus infectious bronchitis virus infection.Virology. 2012 Nov 25;433(2):421-30. doi: 10.1016/j.virol.2012.08.037. Epub 2012 Sep 18. Virology. 2012. PMID: 22995191 Free PMC article.
-
Infectious bronchitis virus E protein is targeted to the Golgi complex and directs release of virus-like particles.J Virol. 2000 May;74(9):4319-26. doi: 10.1128/jvi.74.9.4319-4326.2000. J Virol. 2000. PMID: 10756047 Free PMC article.
-
Coronavirus IBV-induced membrane fusion occurs at near-neutral pH.Arch Virol. 1992;122(3-4):307-16. doi: 10.1007/BF01317192. Arch Virol. 1992. PMID: 1309994 Free PMC article.
Cited by
-
Antigen-Specific Antibody Signature Is Associated with COVID-19 Outcome.Viruses. 2023 Apr 20;15(4):1018. doi: 10.3390/v15041018. Viruses. 2023. PMID: 37112998 Free PMC article.
-
The cellular interactome of the coronavirus infectious bronchitis virus nucleocapsid protein and functional implications for virus biology.J Virol. 2013 Sep;87(17):9486-500. doi: 10.1128/JVI.00321-13. Epub 2013 May 1. J Virol. 2013. PMID: 23637410 Free PMC article.
-
Development of a rapid and sensitive europium (III) chelate microparticle-based lateral flow test strip for the detection and epidemiological surveillance of porcine epidemic diarrhea virus.Arch Virol. 2020 May;165(5):1049-1056. doi: 10.1007/s00705-020-04566-x. Epub 2020 Mar 6. Arch Virol. 2020. PMID: 32144545 Free PMC article.
-
Recombinant viral proteins for use in diagnostic ELISAs to detect virus infection.Vaccine. 2007 Jul 26;25(30):5653-9. doi: 10.1016/j.vaccine.2007.02.053. Epub 2007 Mar 6. Vaccine. 2007. PMID: 17478017 Free PMC article. Review.
-
The coronavirus nucleocapsid is a multifunctional protein.Viruses. 2014 Aug 7;6(8):2991-3018. doi: 10.3390/v6082991. Viruses. 2014. PMID: 25105276 Free PMC article. Review.
References
-
- Alexander D.J., Gouch R.E. A long-term study of the pathogenesis of infections of fowls with three strains of avian infectious bronchitis virus. Res. Vet. Sci. 1978;24:228–233. - PubMed
-
- Ausubel F.M., Brent R., Kingston R.E., Moore D.D., Seidman J.G., Smith J.A., Kevin S. Current Protocols in Molecular Biology. John Wiley and Sons, Inc.; New York: 1987. pp. 10.18.7–10.18.8.
-
- Cheley S., Anderson R. Cellular synthesis and modification of murine hepatitis virus polypeptides. J. Gen. Virol. 1981;54:301–311. - PubMed
-
- Chen H., Gill A., Dove B.K., Emmett S.R., Kemp C.F., Ritchie M.A., Dee M., Hiscox J.A. Mass spectroscopic characterization of the coronavirus infectious bronchitis virus nucleoprotein and elucidation of the role of phosphorylation in RNA binding by using surface plasmon resonance. J. Virol. 2005;79:1164–1179. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

