Central memory self/tumor-reactive CD8+ T cells confer superior antitumor immunity compared with effector memory T cells
- PMID: 15980149
- PMCID: PMC1172264
- DOI: 10.1073/pnas.0503726102
Central memory self/tumor-reactive CD8+ T cells confer superior antitumor immunity compared with effector memory T cells
Abstract
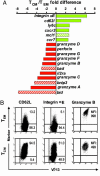
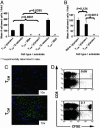
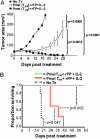
Central memory CD8+ T cells (T(CM)) and effector memory CD8+ T cells (T(EM)) are found in humans and mice; however, their relative contributions to host immunity have only recently been examined in vivo. Further, the ability of T(CM) to treat an established tumor or infection has yet to be evaluated. To address the therapeutic potential of different tumor-reactive CD8+ T cell memory subsets, we used an established model for the in vitro generation of T(CM) and T(EM) by using IL-15 and IL-2, respectively. Adoptively transferred T(CM) exhibited a potent in vivo recall response when combined with tumor-antigen vaccination and exogenous IL-2, leading to the eradication of large established tumors. By contrast, T(EM) were far less effective on a per-cell basis. Microarray analysis revealed that the signature of highly in vivo effective antitumor T cells included the overexpression of genes responsible for trafficking to secondary lymphoid tissues. This gene expression profile correctly predicted the in vitro and in vivo lymphoid-homing attributes of tumor-reactive T cells. Furthermore, we found that homing to secondary lymphoid tissue is required for optimal tumor treatment. Our findings indicated that highly in vivo effective antitumor T cells were those that initially targeted secondary lymphoid tissue, rather than tumor sites, as had previously been postulated. Thus, tumor-reactive CD8+ T cell populations with the phenotypic and functional attributes of T(CM) may be superior to T(EM)/effector T cells for adoptive immunotherapies using concomitant tumor-antigen vaccination.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

