Emergence of a novel mutation in the FLLA region of hepatitis B virus during lamivudine therapy
- PMID: 15980328
- PMCID: PMC1168680
- DOI: 10.1128/AAC.49.7.2618-2624.2005
Emergence of a novel mutation in the FLLA region of hepatitis B virus during lamivudine therapy
Abstract
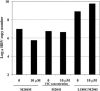
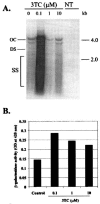
The emergence of resistance to lamivudine has been one of the major stumbling blocks to successful treatment and control of hepatitis B virus (HBV) infections. The major mechanism of resistance has been attributed to the alteration in the YMDD motif of the HBV polymerase due to an amino acid change of rtM204 to V/I and an accompanying rtL180M conversion. A novel mutation pattern in a patient having clinical breakthrough under lamivudine therapy was discovered. The mutant had a rtL180C/M204I genotype and was detected after 2 years of therapy with lamivudine. To characterize this novel variant, site-directed mutagenesis was performed using a vector construct containing the HBV genome. Transient transfection studies in human hepatoma cells with HBV carrying the new mutant demonstrated that the rtL180C/M204I mutant was resistant to lamivudine up to 10 microM. The resistance profile was comparable to that of the previously reported rtL180 M/M204I-containing virus. These observations were further confirmed by generation of stable cultures transfected with the mutant virus.
Figures


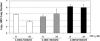

Similar articles
-
Mutational patterns of hepatitis B virus genome and clinical outcomes after emergence of drug-resistant variants during lamivudine therapy: analyses of the polymerase gene and full-length sequences.J Med Virol. 2007 Nov;79(11):1664-70. doi: 10.1002/jmv.20984. J Med Virol. 2007. PMID: 17854034
-
Prevalence and profile of mutations associated with lamivudine therapy in Indian patients with chronic hepatitis B in the surface and polymerase genes of hepatitis B virus.J Med Virol. 2002 Nov;68(3):311-8. doi: 10.1002/jmv.10205. J Med Virol. 2002. PMID: 12226816
-
[YMDD motif variants detected by Inno-Lipa HBV DR assay in chronic hepatitis B patients during lamivudine therapy].Mikrobiyol Bul. 2008 Jul;42(3):445-50. Mikrobiyol Bul. 2008. PMID: 18822888 Turkish.
-
In vitro susceptibility of lamivudine-resistant hepatitis B virus to adefovir and tenofovir.Antivir Ther. 2004 Jun;9(3):353-63. Antivir Ther. 2004. PMID: 15259898
-
Hepatitis B virus resistance to lamivudine and its clinical implications.Antivir Chem Chemother. 2002 May;13(3):143-55. doi: 10.1177/095632020201300301. Antivir Chem Chemother. 2002. PMID: 12448687 Review.
Cited by
-
HBV Drug Resistance Substitutions Existed before the Clinical Approval of Nucleos(t)ide Analogues: A Bioinformatic Analysis by GenBank Data Mining.Viruses. 2017 Jul 27;9(8):199. doi: 10.3390/v9080199. Viruses. 2017. PMID: 28749433 Free PMC article.
-
Emergence of a novel lamivudine-resistant hepatitis B virus variant with a substitution outside the YMDD motif.Antimicrob Agents Chemother. 2006 Nov;50(11):3867-74. doi: 10.1128/AAC.00239-06. Epub 2006 Sep 18. Antimicrob Agents Chemother. 2006. PMID: 16982790 Free PMC article.
References
-
- Allen, M. I., M. Deslauriers, C. W. Andrews, G. A. Tipples, K. A. Walters, D. L. Tyrrell, N. Brown, and L. D. Condreay. 1998. Identification and characterization of mutations in hepatitis B virus resistant to lamivudine. Lamivudine Clinical Investigation Group. Hepatology 27:1670-1677. - PubMed
-
- Angus, P., R. Vaughan, S. Xiong, H. Yang, W. Delaney, C. Gibbs, C. Brosgart, D. Colledge, R. Edwards, A. Ayres, A. Bartholomeusz, and S. Locarnini. 2003. Resistance to adefovir dipivoxil therapy associated with the selection of a novel mutation in the HBV polymerase. Gastroenterology. 125:292-297. - PubMed
-
- Ayres, A., A. Bartholomeusz, G. Lau, K. C. Lam, J. Y. Lee, and S. Locarnini. 2003. Lamivudine and Famciclovir resistant hepatitis B virus associated with fatal hepatic failure. J. Clin. Virol. 27:111-116. - PubMed
-
- Ben-Ari, Z., E. Mor, and R. Tur-Kaspa. 2003. Experience with lamivudine therapy for hepatitis B virus infection before and after liver transplantation, and review of the literature. J. Intern. Med. 253:544-552. - PubMed
-
- Ben-Ari, Z., R. Zemel, A. Kazetsker, G. Fraser, and R. Tur-Kaspa. 1999. Efficacy of lamivudine in patients with hepatitis B virus precore mutant infection before and after liver transplantation. Am. J. Gastroenterol. 94:663-667. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

