Biological activity of an intravenous preparation of human vaccinia immune globulin in mouse models of vaccinia virus infection
- PMID: 15980330
- PMCID: PMC1168682
- DOI: 10.1128/AAC.49.7.2634-2641.2005
Biological activity of an intravenous preparation of human vaccinia immune globulin in mouse models of vaccinia virus infection
Abstract
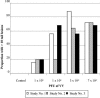
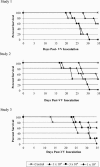
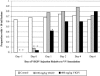
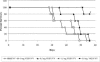
The biological activity of a new intravenous (i.v.) preparation of human vaccinia immune globulin (VIGIV) was evaluated in two mouse models of vaccinia virus (VV) infection. In a mouse tail lesion model, female CD-1 mice were inoculated i.v. with 7 x 10(4) PFU of VV to produce >10 lesions per tail 8 days later. In a mouse lethality model, female severe combined immunodeficient (SCID) mice were inoculated i.v. with 3 x 10(4) PFU of VV to produce 100% mortality within 45 days. The ability of VIGIV to reduce tail lesion formation in CD-1 mice and mortality in SCID mice was determined by (i) pretreatment of a lethal VV dose with VIGIV prior to i.v. inoculation into SCID mice and (ii) i.v. administration of VIGIV to CD-1 and SCID mice the day before and up to 8 days after VV infection. VIGIV reduced the proportion of CD-1 mice with >10 tail lesions in a dose-related manner when VIGIV was given 1 day before and up to 1 day after VV inoculation. The pretreatment of VV with VIGIV prolonged survival and decreased mortality. VIGIV (100 and 400 mg/kg) prolonged survival when given up to 4 days after VV inoculation, and the 400-mg/kg dose reduced the mortality rate by 80% when given the day before or immediately after VV inoculation. The biological activity of VIGIV was demonstrated in both the immunocompetent and immunocompromised murine models. The timing of treatment relative to VV inoculation appeared to be important for the demonstration of VIGIV's biological activity.
Figures
Similar articles
-
Safety and pharmacokinetic evaluation of intravenous vaccinia immune globulin in healthy volunteers.Clin Infect Dis. 2004 Sep 15;39(6):759-66. doi: 10.1086/422998. Epub 2004 Aug 23. Clin Infect Dis. 2004. PMID: 15472804
-
Efficacy of 2-amino-7-(1,3-dihydroxy-2-propoxymethyl)purine for treatment of vaccinia virus (orthopoxvirus) infections in mice.Antimicrob Agents Chemother. 2001 Jan;45(1):84-7. doi: 10.1128/AAC.45.1.84-87.2001. Antimicrob Agents Chemother. 2001. PMID: 11120949 Free PMC article.
-
Administration of vaccinia virus to mice may cause contact or bedding sentinel mice to test positive for orthopoxvirus antibodies: case report and follow-up investigation.Comp Med. 2003 Feb;53(1):85-8. Comp Med. 2003. PMID: 12625512
-
Effect of 5-iodo-2'-deoxyuridine on vaccinia virus (orthopoxvirus) infections in mice.Antimicrob Agents Chemother. 2002 Sep;46(9):2842-7. doi: 10.1128/AAC.46.9.2842-2847.2002. Antimicrob Agents Chemother. 2002. PMID: 12183236 Free PMC article.
-
Vaccinia immune globulin: current policies, preparedness, and product safety and efficacy.Int J Infect Dis. 2006 May;10(3):193-201. doi: 10.1016/j.ijid.2005.12.001. Epub 2006 Mar 27. Int J Infect Dis. 2006. PMID: 16564720 Review.
Cited by
-
African Swine Fever Vaccinology: The Biological Challenges from Immunological Perspectives.Viruses. 2022 Sep 13;14(9):2021. doi: 10.3390/v14092021. Viruses. 2022. PMID: 36146827 Free PMC article. Review.
-
Enhanced efficacy of cidofovir combined with vaccinia immune globulin in treating progressive cutaneous vaccinia virus infections in immunosuppressed hairless mice.Antimicrob Agents Chemother. 2015 Jan;59(1):520-6. doi: 10.1128/AAC.04289-14. Epub 2014 Nov 10. Antimicrob Agents Chemother. 2015. PMID: 25385098 Free PMC article.
-
Protection of rabbits and immunodeficient mice against lethal poxvirus infections by human monoclonal antibodies.PLoS One. 2012;7(11):e48706. doi: 10.1371/journal.pone.0048706. Epub 2012 Nov 2. PLoS One. 2012. PMID: 23133652 Free PMC article.
-
Mpox (formerly monkeypox): pathogenesis, prevention, and treatment.Signal Transduct Target Ther. 2023 Dec 27;8(1):458. doi: 10.1038/s41392-023-01675-2. Signal Transduct Target Ther. 2023. PMID: 38148355 Free PMC article. Review.
-
Paediatric, maternal, and congenital mpox: a systematic review and meta-analysis.Lancet Glob Health. 2024 Apr;12(4):e572-e588. doi: 10.1016/S2214-109X(23)00607-1. Epub 2024 Feb 21. Lancet Glob Health. 2024. PMID: 38401556 Free PMC article.
References
-
- Belyakov, I. M., P. Earl, A. Dzutsev, V. A. Kuznetsov, M. Lemon, L. S. Wyatt, J. T. Snyder, J. D. Ahlers, G. Franchini, B. Moss, and J. A. Berzofsky. 2003. Shared modes of protection against poxvirus infection by attenuated and conventional smallpox vaccine viruses. Proc. Natl. Acad. Sci. USA 100:9458-9463. - PMC - PubMed
-
- Boyle, J. J., R. F. Haff, and R. C. Stewart. 1966. Evaluation of antiviral compounds by suppression of tail lesions in vaccinia-infected mice. Antimicrob. Agents Chemother. 6:536-539. - PubMed
-
- Bray, M. 2003. Pathogenesis and potential antiviral therapy of complications of smallpox vaccination. Antiviral Res. 58:101-114. - PubMed
-
- Bray, M., and M. E. Wright. 2003. Progressive vaccinia. Clin. Infect. Dis. 36:766-774. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

