A model of the ternary complex of interleukin-10 with its soluble receptors
- PMID: 15985167
- PMCID: PMC1192808
- DOI: 10.1186/1472-6807-5-10
A model of the ternary complex of interleukin-10 with its soluble receptors
Abstract
Background: Interleukin-10 (IL-10) is a cytokine whose main biological function is to suppress the immune response by induction of a signal(s) leading to inhibition of synthesis of a number of cytokines and their cellular receptors. Signal transduction is initiated upon formation of a ternary complex of IL-10 with two of its receptor chains, IL-10R1 and IL-10R2, expressed on the cell membrane. The affinity of IL-10R1 toward IL-10 is very high, which allowed determination of the crystal structure of IL-10 complexed with the extracellular/soluble domain of IL-10R1, while the affinity of IL-10R2 toward either IL-10 or IL-10/sIL-10R1 complex is quite low. This so far has prevented any attempts to obtain structural information about the ternary complex of IL-10 with its receptor chains.
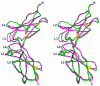
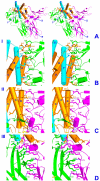
Results: Structures of the second soluble receptor chain of interleukin-10 (sIL-10R2) and the ternary complex of IL-10/sIL-10R1/sIL-10R2 have been generated by homology modeling, which allowed us to identify residues involved in ligand-receptor and receptor-receptor interactions.
Conclusion: The previously experimentally determined structure of the intermediate/binary complex IL-10/sIL-10R1 is the same in the ternary complex. There are two binding sites for the second receptor chain on the surface of the IL-10/sIL-10R1 complex, involving both IL-10 and sIL-10R1. Most of the interactions are hydrophilic in nature, although each interface includes two internal hydrophobic clusters. The distance between C-termini of the receptor chains is 25 A, which is common for known structures of ternary complexes of other cytokines. The structure is likely to represent the biologically active signaling complex of IL-10 with its receptor on the surface of the cell membrane.
Figures
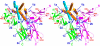
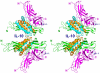
Similar articles
-
Crystal structure of the IL-10/IL-10R1 complex reveals a shared receptor binding site.Immunity. 2001 Jul;15(1):35-46. doi: 10.1016/s1074-7613(01)00169-8. Immunity. 2001. PMID: 11485736
-
Comparison of interleukin-22 and interleukin-10 soluble receptor complexes.J Interferon Cytokine Res. 2002 Nov;22(11):1099-112. doi: 10.1089/10799900260442520. J Interferon Cytokine Res. 2002. PMID: 12513909
-
Crystal structure of human cytomegalovirus IL-10 bound to soluble human IL-10R1.Proc Natl Acad Sci U S A. 2002 Jul 9;99(14):9404-9. doi: 10.1073/pnas.152147499. Epub 2002 Jul 1. Proc Natl Acad Sci U S A. 2002. PMID: 12093920 Free PMC article.
-
Structural analysis of cytokines comprising the IL-10 family.Cytokine Growth Factor Rev. 2010 Oct;21(5):325-30. doi: 10.1016/j.cytogfr.2010.08.003. Epub 2010 Sep 16. Cytokine Growth Factor Rev. 2010. PMID: 20846897 Free PMC article. Review.
-
Structural features of the interleukin-10 family of cytokines.Curr Pharm Des. 2004;10(31):3873-84. doi: 10.2174/1381612043382602. Curr Pharm Des. 2004. PMID: 15579076 Review.
Cited by
-
Parapoxvirus Interleukin-10 Homologues Vary in Their Receptor Binding, Anti-Inflammatory, and Stimulatory Activities.Pathogens. 2022 Apr 24;11(5):507. doi: 10.3390/pathogens11050507. Pathogens. 2022. PMID: 35631028 Free PMC article.
-
Crystal structure of human interferon-λ1 in complex with its high-affinity receptor interferon-λR1.J Mol Biol. 2010 Dec 10;404(4):650-64. doi: 10.1016/j.jmb.2010.09.068. Epub 2010 Oct 8. J Mol Biol. 2010. PMID: 20934432 Free PMC article.
-
Blocking IL-10 signaling with soluble IL-10 receptor restores in vitro specific lymphoproliferative response in dogs with leishmaniasis caused by Leishmania infantum.PLoS One. 2021 Jan 19;16(1):e0239171. doi: 10.1371/journal.pone.0239171. eCollection 2021. PLoS One. 2021. PMID: 33465107 Free PMC article.
-
Koi herpesvirus encodes and expresses a functional interleukin-10.J Virol. 2012 Nov;86(21):11512-20. doi: 10.1128/JVI.00957-12. Epub 2012 Aug 15. J Virol. 2012. PMID: 22896613 Free PMC article.
-
Novel exonic mutation inducing aberrant splicing in the IL10RA gene and resulting in infantile-onset inflammatory bowel disease: a case report.BMC Gastroenterol. 2016 Jan 28;16:10. doi: 10.1186/s12876-016-0424-5. BMC Gastroenterol. 2016. PMID: 26822028 Free PMC article.
References
-
- D'Andrea A, Aste-Amezaga M, Valiante NM, Ma X, Kubin M, Trinchieri G. Interleukin 10 (IL-10) inhibits human lymphocyte interferon gamma-production by suppressing natural killer cell stimulatory factor/IL-12 synthesis in accessory cells. J Exp Med. 1993;178:1041–1048. doi: 10.1084/jem.178.3.1041. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

