Glucosylation of beta-lactoglobulin lowers the heat capacity change of unfolding; a unique way to affect protein thermodynamics
- PMID: 15987887
- PMCID: PMC2279330
- DOI: 10.1110/ps.051405005
Glucosylation of beta-lactoglobulin lowers the heat capacity change of unfolding; a unique way to affect protein thermodynamics
Abstract
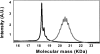
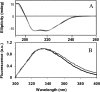
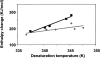
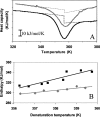
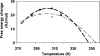
Chemical glycosylation of proteins occurs in vivo spontaneously, especially under stress conditions, and has been linked in a number of cases to diseases related to protein denaturation and aggregation. It is the aim of this work to study the origin of the change in thermodynamic properties due to glucosylation of the folded beta-lactoglobulin A. Under mild conditions Maillard products can be formed by reaction of epsilon-amino groups of lysines with the reducing group of, in this case, glucose. The formed conjugates described here have an average degree of glycosylation of 82%. No impact of the glucosylation on the protein structure is detected, except that the Stokes radius was increased by approximately 3%. Although at ambient temperatures the change in Gibbs energy of unfolding is reduced by 20%, the denaturation temperature is increased by 5 degrees C. Using a combination of circular dichroism, fluorescence, and calorimetric approaches, it is shown that the change in heat capacity upon denaturation is reduced by 60% due to the glucosylation. Since in the denatured state the Stokes radius of the protein is not significantly smaller for the glucosylated protein, it is suggested that the nonpolar residues associate to the covalently linked sugar moiety in the unfolded state, thereby preventing their solvent exposure. In this way coupling of small reducing sugar moieties to solvent exposed groups of proteins offers an efficient and unique tool to deal with protein stability issues, relevant not only in nature but also for technological applications.
Figures


Similar articles
-
Controlling the aggregation propensity and thereby digestibility of allergens by Maillardation as illustrated for cod fish parvalbumin.J Biosci Bioeng. 2011 Feb;111(2):204-11. doi: 10.1016/j.jbiosc.2010.09.015. Epub 2010 Oct 20. J Biosci Bioeng. 2011. PMID: 20970374
-
Identification of pitfalls in the analysis of heat capacity changes of beta-lactoglobulin A.Int J Biol Macromol. 2005 Oct 30;37(1-2):28-34. doi: 10.1016/j.ijbiomac.2005.08.001. Epub 2005 Sep 28. Int J Biol Macromol. 2005. PMID: 16197991
-
[A comparative thermodynamic study of heat and cold denaturation of beta-lactoglobulin].Mol Biol (Mosk). 1992 Mar-Apr;26(2):285-91. Mol Biol (Mosk). 1992. PMID: 1339949 Russian.
-
Impact of Maillard type glycation on properties of beta-lactoglobulin.Biotechnol Adv. 2006 Nov-Dec;24(6):629-32. doi: 10.1016/j.biotechadv.2006.07.004. Epub 2006 Jul 8. Biotechnol Adv. 2006. PMID: 16904283 Review.
-
Thermal unfolding of beta-lactoglobulin, alpha-lactalbumin, and bovine serum albumin. A thermodynamic approach.Crit Rev Food Sci Nutr. 1996 Jul;36(6):565-601. doi: 10.1080/10408399609527740. Crit Rev Food Sci Nutr. 1996. PMID: 8841732 Review.
Cited by
-
Transglutaminase-Catalyzed Glycosylation Improved Physicochemical and Functional Properties of Lentinus edodes Protein Fraction.Foods. 2023 Apr 29;12(9):1849. doi: 10.3390/foods12091849. Foods. 2023. PMID: 37174388 Free PMC article.
-
Preparation and Evaluation of New Glycopeptides Obtained by Proteolysis from Corn Gluten Meal Followed by Transglutaminase-Induced Glycosylation with Glucosamine.Foods. 2020 May 1;9(5):555. doi: 10.3390/foods9050555. Foods. 2020. PMID: 32370047 Free PMC article.
-
Milk Processing Affects Structure, Bioavailability and Immunogenicity of β-lactoglobulin.Foods. 2020 Jul 3;9(7):874. doi: 10.3390/foods9070874. Foods. 2020. PMID: 32635246 Free PMC article. Review.
-
Digestibility and IgE-binding of glycosylated codfish parvalbumin.Biomed Res Int. 2013;2013:756789. doi: 10.1155/2013/756789. Epub 2013 Jun 26. Biomed Res Int. 2013. PMID: 23878817 Free PMC article.
-
Effect of quality of milk on maillard reaction and protein oxidation during preparation of cow and buffalo milk khoa.J Food Sci Technol. 2017 Aug;54(9):2737-2745. doi: 10.1007/s13197-017-2710-9. Epub 2017 May 23. J Food Sci Technol. 2017. PMID: 28928513 Free PMC article.
References
-
- Broersen, K., Voragen, A.G.J., Hamer, R.J., and de Jongh, H.H.J. 2004. Glycoforms of β-lactoglobulin with improved thermostability and preserved structural packing. Biotech. Bioeng. 86 78–87. - PubMed
-
- de Jongh, H.H.J., Gröneveld, T., and de Groot, J. 2001. Mild isolation procedure discloses new protein structural properties of β-lactoglobulin. J. Dairy Sci. 84 562–571. - PubMed
-
- Digris, A.V., Skakun, V.V., Novikov, E.G., van Hoek, A., Claiborne, A., and Visser, A.J.W.G. 1999. Thermal stability of a flavoprotein assessed from associative analysis of polarized time-resolved fluorescence spectroscopy. Eur. Biophys. J. 28 526–531. - PubMed
-
- Endo, Y., Nagai, H., Watanabe, Y., Ochi, K., and Takagi, T. 1992. Heat-induced aggregation of recombinant erythropoietin in the intact and deglycosylated states as monitored by gel-permeation chromatography combined with a low-angle laser-light scattering technique. J. Biochem. (Tokyo) 112 700–706. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

