Apical entry and release of severe acute respiratory syndrome-associated coronavirus in polarized Calu-3 lung epithelial cells
- PMID: 16014910
- PMCID: PMC1181546
- DOI: 10.1128/JVI.79.15.9470-9479.2005
Apical entry and release of severe acute respiratory syndrome-associated coronavirus in polarized Calu-3 lung epithelial cells
Abstract
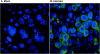
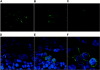
Severe acute respiratory syndrome (SARS), caused by a novel coronavirus (CoV) known as SARS-CoV, is a contagious and life-threatening respiratory illness with pneumocytes as its main target. A full understanding of how SARS-CoV would interact with lung epithelial cells will be vital for advancing our knowledge of SARS pathogenesis. However, an in vitro model of SARS-CoV infection using relevant lung epithelial cells is not yet available, making it difficult to dissect the pathogenesis of SARS-CoV in the lungs. Here, we report that SARS-CoV can productively infect human bronchial epithelial Calu-3 cells, causing cytopathic effects, a process reflective of its natural course of infection in the lungs. Indirect immunofluorescence studies revealed a preferential expression of angiotensin-converting enzyme 2 (ACE-2), the functional receptor of SARS-CoV, on the apical surface. Importantly, both ACE-2 and viral antigen appeared to preferentially colocalize at the apical domain of infected cells. In highly polarized Calu-3 cells grown on the membrane inserts, we found that cells exposed to virus through the apical rather than the basolateral surface showed high levels of viral replication. Progeny virus was released into the apical chamber at titers up to 5 logs higher than those recovered from the basolateral chambers of polarized cultures. Taken together, these results indicate that SARS-CoV almost exclusively entered and was released from the apical domain of polarized Calu-3 cells, which might provide important insight into the mechanism of transmission and pathogenesis of SARS-CoV.
Figures




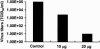


Similar articles
-
SARS-CoV replication and pathogenesis in an in vitro model of the human conducting airway epithelium.Virus Res. 2008 Apr;133(1):33-44. doi: 10.1016/j.virusres.2007.03.013. Epub 2007 Apr 23. Virus Res. 2008. PMID: 17451829 Free PMC article. Review.
-
ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia.J Virol. 2005 Dec;79(23):14614-21. doi: 10.1128/JVI.79.23.14614-14621.2005. J Virol. 2005. PMID: 16282461 Free PMC article.
-
Analysis of ACE2 in polarized epithelial cells: surface expression and function as receptor for severe acute respiratory syndrome-associated coronavirus.J Gen Virol. 2006 Jun;87(Pt 6):1691-1695. doi: 10.1099/vir.0.81749-0. J Gen Virol. 2006. PMID: 16690935
-
Bilateral entry and release of Middle East respiratory syndrome coronavirus induces profound apoptosis of human bronchial epithelial cells.J Virol. 2013 Sep;87(17):9953-8. doi: 10.1128/JVI.01562-13. Epub 2013 Jul 3. J Virol. 2013. PMID: 23824802 Free PMC article.
-
Receptor recognition and cross-species infections of SARS coronavirus.Antiviral Res. 2013 Oct;100(1):246-54. doi: 10.1016/j.antiviral.2013.08.014. Epub 2013 Aug 29. Antiviral Res. 2013. PMID: 23994189 Free PMC article. Review.
Cited by
-
SARS-CoV-2: Pathogenesis, Molecular Targets and Experimental Models.Front Pharmacol. 2021 Apr 22;12:638334. doi: 10.3389/fphar.2021.638334. eCollection 2021. Front Pharmacol. 2021. PMID: 33967772 Free PMC article. Review.
-
Porcine aminopeptidase N mediated polarized infection by porcine epidemic diarrhea virus in target cells.Virology. 2015 Apr;478:1-8. doi: 10.1016/j.virol.2015.01.020. Epub 2015 Feb 11. Virology. 2015. PMID: 25681796 Free PMC article.
-
Generation of a transgenic mouse model of Middle East respiratory syndrome coronavirus infection and disease.J Virol. 2015 Apr;89(7):3659-70. doi: 10.1128/JVI.03427-14. Epub 2015 Jan 14. J Virol. 2015. PMID: 25589660 Free PMC article.
-
SARS-CoV-2: Ultrastructural Characterization of Morphogenesis in an In Vitro System.Viruses. 2022 Jan 20;14(2):201. doi: 10.3390/v14020201. Viruses. 2022. PMID: 35215794 Free PMC article.
-
Emerging SARS-CoV-2 Genotypes Show Different Replication Patterns in Human Pulmonary and Intestinal Epithelial Cells.Viruses. 2021 Dec 23;14(1):23. doi: 10.3390/v14010023. Viruses. 2021. PMID: 35062227 Free PMC article.
References
-
- Basbaum, C. B., J. M. Madison, C. P. Sommerhoff, J. K. Brown, and W. E. Finkbeiner. 1990. Receptors on airway gland cells. Am. Rev. Respir. Dis. 141:S141-S144. - PubMed
-
- Blau, D. M., and R. W. Compans. 1995. Entry and release of measles virus are polarized in epithelial cells. Virology 210:91-99. - PubMed
-
- Centers for Disease Control and Prevention. 2003. Outbreak of severe acute respiratory syndrome—worldwide, 2003. Morb. Mortal. Wkly. Rep. 52:226-228. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

