Lack of NMDA receptor subtype selectivity for hippocampal long-term potentiation
- PMID: 16033900
- PMCID: PMC6725356
- DOI: 10.1523/JNEUROSCI.1905-05.2005
Lack of NMDA receptor subtype selectivity for hippocampal long-term potentiation
Abstract
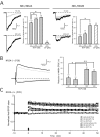
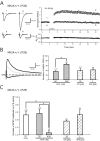
NMDA receptor (NMDAR) 2A (NR2A)- and NR2B-type NMDARs coexist in synapses of CA1 pyramidal cells. Recent studies using pharmacological blockade of NMDAR subtypes proposed that the NR2A type is responsible for inducing long-term potentiation (LTP), whereas the NR2B type induces long-term depression (LTD). This contrasts with the finding in genetically modified mice that NR2B-type NMDARs induce LTP when NR2A signaling is absent or impaired, although compensatory mechanisms might have contributed to this result. We therefore assessed the contribution of the two NMDAR subtypes to LTP in mouse hippocampal slices by different induction protocols and in the presence of NMDAR antagonists, including the NR2A-type blocker NVP-AAM077, for which an optimal concentration for subtype selectivity was determined on recombinant and native NMDARs. Partial blockade of NMDA EPSCs by 40%, either by preferentially antagonizing NR2A- or NR2B-type NMDARs or by the nonselective antagonist D-AP-5, did not impair LTP, demonstrating that hippocampal LTP induction can be generated by either NMDAR subtype.
Figures
References
-
- Auberson YP, Allgeier H, Bischoff S, Lingenhoehl K, Moretti R, Schmutz M (2002) 5-Phosphonomethylquinoxalinediones as competitive NMDA receptor antagonists with a preference for the human 1A/2A, rather than 1A/2B receptor composition. Bioorg Med Chem Lett 12: 1099-1102. - PubMed
-
- Collingridge GL, Isaac JTR, Wang YT (2004) Receptor trafficking and synaptic plasticity. Nat Rev Neurosci 5: 952-962. - PubMed
-
- Cummings JA, Mulkey RM, Nicoll RA, Malenka RC (1996) Ca2+ signaling requirements for long-term depression in the hippocampus. Neuron 16: 825-833. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
