DNA-based vaccines activate innate and adaptive antitumor immunity by engaging the NKG2D receptor
- PMID: 16040807
- PMCID: PMC1182421
- DOI: 10.1073/pnas.0502208102
DNA-based vaccines activate innate and adaptive antitumor immunity by engaging the NKG2D receptor
Abstract
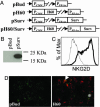
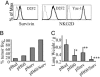
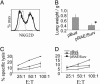
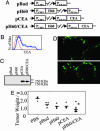
The interaction of NKG2D, a stimulatory receptor expressed on natural killer (NK) cells and activated CD8(+) T cells, and its ligands mediates stimulatory and costimulatory signals to these cells. Here, we demonstrate that DNA-based vaccines, encoding syngeneic or allogeneic NKG2D ligands together with tumor antigens such as survivin or carcinoembryonic antigen, markedly activate both innate and adaptive antitumor immunity. Such vaccines result in highly effective, NK- and CD8(+) T cell-mediated protection against either breast or colon carcinoma cells in prophylactic and therapeutic settings. Notably, this protection was irrespective of the NKG2D ligand expression level of the tumor cells. Hence, this strategy has the potential to lead to widely applicable and possibly clinically useful DNA-based cancer vaccines.
Figures



Similar articles
-
A DNA-based cancer vaccine enhances lymphocyte cross talk by engaging the NKG2D receptor.Blood. 2006 Apr 15;107(8):3251-7. doi: 10.1182/blood-2005-10-4231. Epub 2005 Dec 20. Blood. 2006. PMID: 16368879 Free PMC article.
-
Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity.Nature. 2001 Sep 13;413(6852):165-71. doi: 10.1038/35093109. Nature. 2001. PMID: 11557981 Free PMC article.
-
Ligands for the murine NKG2D receptor: expression by tumor cells and activation of NK cells and macrophages.Nat Immunol. 2000 Aug;1(2):119-26. doi: 10.1038/77793. Nat Immunol. 2000. PMID: 11248803
-
Strategies for target cell recognition by natural killer cells.Immunol Rev. 2001 Jun;181:170-84. doi: 10.1034/j.1600-065x.2001.1810114.x. Immunol Rev. 2001. PMID: 11513138 Review.
-
NKG2D and Related Immunoreceptors.Adv Protein Chem. 2004;68:281-312. doi: 10.1016/S0065-3233(04)68008-9. Adv Protein Chem. 2004. PMID: 15500864 Review.
Cited by
-
An epigenetic vaccine model active in the prevention and treatment of melanoma.J Transl Med. 2007 Dec 10;5:64. doi: 10.1186/1479-5876-5-64. J Transl Med. 2007. PMID: 18070359 Free PMC article.
-
A Legumain-based minigene vaccine targets the tumor stroma and suppresses breast cancer growth and angiogenesis.Cancer Immunol Immunother. 2008 Apr;57(4):507-15. doi: 10.1007/s00262-007-0389-x. Epub 2007 Sep 5. Cancer Immunol Immunother. 2008. PMID: 17786443 Free PMC article.
-
Oncogenic stress sensed by the immune system: role of natural killer cell receptors.Nat Rev Immunol. 2009 Aug;9(8):568-80. doi: 10.1038/nri2604. Nat Rev Immunol. 2009. PMID: 19629084 Free PMC article. Review.
-
Improved binding activity of antibodies against major histocompatibility complex class I chain-related gene A by phage display technology for cancer-targeted therapy.J Biomed Biotechnol. 2012;2012:597647. doi: 10.1155/2012/597647. Epub 2012 Nov 21. J Biomed Biotechnol. 2012. PMID: 23226940 Free PMC article.
-
Changes to the natural killer cell repertoire after therapeutic hepatitis B DNA vaccination.PLoS One. 2010 Jan 18;5(1):e8761. doi: 10.1371/journal.pone.0008761. PLoS One. 2010. PMID: 20090916 Free PMC article. Clinical Trial.
References
-
- Diefenbach, A. & Raulet, D. H. (2002) Immunol. Rev. 188, 9-21. - PubMed
-
- Diefenbach, A., Jamieson, A. M., Liu, S. D., Shastri, N. & Raulet, D. H. (2000) Nat. Immunol. 1, 119-126. - PubMed
-
- Jamieson, A. M., Diefenbach, A., McMahon, C. W., Xiong, N., Carlyle, J. R. & Raulet, D. H. (2002) Immunity 17, 19-29. - PubMed
-
- Snyder, M. R., Weyand, C. M. & Goronzy, J. J. (2004) Trends Immunol. 25, 25-32. - PubMed
-
- Cerwenka, A., Bakker, A. B., McClanahan, T., Wagner, J., Wu, J., Phillips, J. H. & Lanier, L. L. (2000) Immunity 12, 721-727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

