Formation of morphologically similar globular aggregates from diverse aggregation-prone proteins in mammalian cells
- PMID: 16040812
- PMCID: PMC1182404
- DOI: 10.1073/pnas.0409283102
Formation of morphologically similar globular aggregates from diverse aggregation-prone proteins in mammalian cells
Abstract
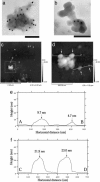
Huntington's disease is a progressive neurodegenerative disorder caused by a polyglutamine repeat expansion in the first exon of the huntingtin (Htt) protein. N-terminal Htt peptides with polyglutamine tracts in the pathological range (51-122 glutamines) form high-molecular-weight protein aggregates with fibrillar morphology in vitro, and they form discrete inclusion bodies in a cell-culture model. However, in some studies, formation of discrete Htt inclusions does not correlate well with cell death. We coexpressed N-terminal Htt fragments containing 91 glutamines fused to different affinity tags in HEK293 cells, and we isolated small aggregates by double sequential-affinity chromatography to assure the isolation of multimeric molecules. Transmission electron microscopy and atomic force microscopy revealed the isolated aggregates as globules or clusters of globules 4-50 nm in diameter without any detectable fibrillar species. Because small nonfibrillar oligomers, not mature fibrils, recently have been suggested to be the principal cytotoxic species in neurodegenerative disease, these Htt globular aggregates formed in cells may represent the pathogenic form of mutant Htt.
Figures


Similar articles
-
Mutant huntingtin promotes the fibrillogenesis of wild-type huntingtin: a potential mechanism for loss of huntingtin function in Huntington's disease.J Biol Chem. 2003 Oct 17;278(42):41452-61. doi: 10.1074/jbc.M303354200. Epub 2003 Jul 29. J Biol Chem. 2003. PMID: 12888569
-
Huntingtin spheroids and protofibrils as precursors in polyglutamine fibrilization.J Biol Chem. 2002 Oct 25;277(43):41032-7. doi: 10.1074/jbc.M205809200. Epub 2002 Aug 8. J Biol Chem. 2002. PMID: 12171927
-
Atomic force microscopy analysis of the Huntington protein nanofibril formation.Nanomedicine. 2005 Mar;1(1):52-7. doi: 10.1016/j.nano.2004.11.004. Nanomedicine. 2005. PMID: 17292058
-
Assessing mutant huntingtin fragment and polyglutamine aggregation by atomic force microscopy.Methods. 2011 Mar;53(3):275-84. doi: 10.1016/j.ymeth.2010.12.028. Epub 2010 Dec 25. Methods. 2011. PMID: 21187152
-
Mutant huntingtin fragments form oligomers in a polyglutamine length-dependent manner in vitro and in vivo.J Biol Chem. 2010 May 7;285(19):14777-90. doi: 10.1074/jbc.M109.093708. Epub 2010 Mar 10. J Biol Chem. 2010. PMID: 20220138 Free PMC article.
Cited by
-
Expression of the type VI intermediate filament proteins CP49 and filensin in the mouse lens epithelium.Mol Vis. 2016 Aug 6;22:970-89. eCollection 2016. Mol Vis. 2016. PMID: 27559293 Free PMC article.
-
Protein quality control during erythropoiesis and hemoglobin synthesis.Hematol Oncol Clin North Am. 2010 Dec;24(6):1071-88. doi: 10.1016/j.hoc.2010.08.013. Hematol Oncol Clin North Am. 2010. PMID: 21075281 Free PMC article. Review.
-
Atomistic simulations of the effects of polyglutamine chain length and solvent quality on conformational equilibria and spontaneous homodimerization.J Mol Biol. 2008 Dec 5;384(1):279-97. doi: 10.1016/j.jmb.2008.09.026. Epub 2008 Sep 18. J Mol Biol. 2008. PMID: 18824003 Free PMC article.
-
Tadpole-like Conformations of Huntingtin Exon 1 Are Characterized by Conformational Heterogeneity that Persists regardless of Polyglutamine Length.J Mol Biol. 2018 May 11;430(10):1442-1458. doi: 10.1016/j.jmb.2018.03.031. Epub 2018 Apr 5. J Mol Biol. 2018. PMID: 29627459 Free PMC article.
-
Sedimentation Velocity Analysis with Fluorescence Detection of Mutant Huntingtin Exon 1 Aggregation in Drosophila melanogaster and Caenorhabditis elegans.Biochemistry. 2017 Sep 5;56(35):4676-4688. doi: 10.1021/acs.biochem.7b00518. Epub 2017 Aug 24. Biochemistry. 2017. PMID: 28786671 Free PMC article.
References
-
- Harper, P. (1996) Huntington's Disease (Saunders, Philadelphia).
-
- Vonsattel, J. P., Myers, R. H., Stevens, T. J., Ferrante, R. J., Bird, E. D. & Richardson, E. P., Jr., (1985) J. Neuropathol. Exp. Neurol. 44, 559-577. - PubMed
-
- The Huntington's Disease Collaborative Research Group (1993) Cell 72, 971-983. - PubMed
-
- Cooper, J. K., Schilling, G., Peters, M. F., Herring, W. J., Sharp, A. H., Kaminsky, Z., Masone, J., Khan, F. A., Delanoy, M., Borchelt, D. R., et al. (1998) Hum. Mol. Genet. 7, 783-790. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

