HIV-1 Nef down-regulates the hemochromatosis protein HFE, manipulating cellular iron homeostasis
- PMID: 16043695
- PMCID: PMC1180511
- DOI: 10.1073/pnas.0504823102
HIV-1 Nef down-regulates the hemochromatosis protein HFE, manipulating cellular iron homeostasis
Abstract
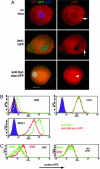
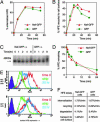
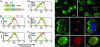
The multifunctional Nef protein of HIV-1 is important for the progression to AIDS. One action of Nef is to down-regulate surface MHC I molecules, helping infected cells to evade immunity. We found that Nef also down-regulates the macrophage-expressed MHC 1b protein HFE, which regulates iron homeostasis and is mutated in the iron-overloading disorder hemochromatosis. In model cell lines, Nef reroutes HFE to a perinuclear structure that overlaps the trans-Golgi network, causing a 90% reduction of surface HFE. This activity requires a Src-kinase-binding proline-rich domain of Nef and a conserved tyrosine-based motif in the cytoplasmic tail of HFE. HIV-1 infection of ex vivo macrophages similarly down-regulates naturally expressed surface HFE in a Nef-dependent manner. The effect of Nef expression on cellular iron was explored; iron and ferritin accumulation were increased in HIV-1-infected ex vivo macrophages expressing wild-type HFE, but this effect was lost with Nef-deleted HIV-1 or when infecting macrophages from hemochromatosis patients expressing mutated HFE. The iron accumulation in HIV-1-infected HFE-expressing macrophages was paralleled by an increase in cellular HIV-1-gag expression. We conclude that, through Nef and HFE, HIV-1 directly regulates cellular iron metabolism, possibly benefiting viral growth.
Figures



Similar articles
-
Wild-type HFE protein normalizes transferrin iron accumulation in macrophages from subjects with hereditary hemochromatosis.Blood. 2000 Aug 1;96(3):1125-9. Blood. 2000. PMID: 10910932
-
Simian and human immunodeficiency virus Nef proteins use different surfaces to downregulate class I major histocompatibility complex antigen expression.J Virol. 2000 Jun;74(12):5691-701. doi: 10.1128/jvi.74.12.5691-5701.2000. J Virol. 2000. PMID: 10823877 Free PMC article.
-
The hemochromatosis protein HFE inhibits iron export from macrophages.Proc Natl Acad Sci U S A. 2002 Nov 26;99(24):15602-7. doi: 10.1073/pnas.242614699. Epub 2002 Nov 12. Proc Natl Acad Sci U S A. 2002. PMID: 12429850 Free PMC article.
-
HFE, the MHC and hemochromatosis: paradigm for an extended function for MHC class I.Tissue Antigens. 2003 Apr;61(4):263-75. doi: 10.1034/j.1399-0039.2003.00065.x. Tissue Antigens. 2003. PMID: 12753664 Review.
-
Possible roles of the hereditary hemochromatosis protein, HFE, in regulating cellular iron homeostasis.Biol Res. 2006;39(1):105-11. doi: 10.4067/s0716-97602006000100013. Biol Res. 2006. PMID: 16629171 Review.
Cited by
-
Cerebrospinal fluid (CSF) biomarkers of iron status are associated with CSF viral load, antiretroviral therapy, and demographic factors in HIV-infected adults.Fluids Barriers CNS. 2017 Apr 21;14(1):11. doi: 10.1186/s12987-017-0058-1. Fluids Barriers CNS. 2017. PMID: 28427421 Free PMC article.
-
Crossing the Iron Gate: Why and How Transferrin Receptors Mediate Viral Entry.Annu Rev Nutr. 2018 Aug 21;38:431-458. doi: 10.1146/annurev-nutr-082117-051749. Epub 2018 May 31. Annu Rev Nutr. 2018. PMID: 29852086 Free PMC article. Review.
-
Intracellular Low Iron Exerts Anti-BK Polyomavirus Effect by Inhibiting the Protein Synthesis of Exogenous Genes.Microbiol Spectr. 2021 Dec 22;9(3):e0109421. doi: 10.1128/Spectrum.01094-21. Epub 2021 Nov 10. Microbiol Spectr. 2021. PMID: 34756078 Free PMC article.
-
Inverse relationship of serum hepcidin levels with CD4 cell counts in HIV-infected patients selected from an Indonesian prospective cohort study.PLoS One. 2013 Nov 11;8(11):e79904. doi: 10.1371/journal.pone.0079904. eCollection 2013. PLoS One. 2013. PMID: 24244576 Free PMC article.
-
Anemia and iron homeostasis in a cohort of HIV-infected patients in Indonesia.BMC Infect Dis. 2011 Aug 9;11:213. doi: 10.1186/1471-2334-11-213. BMC Infect Dis. 2011. PMID: 21827653 Free PMC article.
References
-
- Deacon, N. J., Tsykin, A., Solomon, A., Smith, K., Ludford-Menting, M., Hooker, D. J., McPhee, D. A., Greenway, A. L., Ellett, A., Chatfield, C., et al. (1995) Science 270, 988-991. - PubMed
-
- She, H., Xiong, S., Lin, M., Zandi, E., Giulivi, C. & Tsukamoto, H. (2002) in Am. J. Physiol. 283, G719-G726. - PubMed
-
- Guy, B., Kieny, M. P., Riviere, Y., Le Peuch, C., Dott, K., Girard, M., Montagnier, L. & Lecocq, J. P. (1987) Nature 330, 266-269. - PubMed
-
- Aiken, C., Konner, J., Landau, N. R., Lenburg, M. E. & Trono, D. (1994) Cell 76, 853-864. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

