Dephosphorylation of CDK9 by protein phosphatase 2A and protein phosphatase-1 in Tat-activated HIV-1 transcription
- PMID: 16048649
- PMCID: PMC1187922
- DOI: 10.1186/1742-4690-2-47
Dephosphorylation of CDK9 by protein phosphatase 2A and protein phosphatase-1 in Tat-activated HIV-1 transcription
Abstract
Background: HIV-1 Tat protein recruits human positive transcription elongation factor P-TEFb, consisting of CDK9 and cyclin T1, to HIV-1 transactivation response (TAR) RNA. CDK9 is maintained in dephosphorylated state by TFIIH and undergo phosphorylation upon the dissociation of TFIIH. Thus, dephosphorylation of CDK9 prior to its association with HIV-1 preinitiation complex might be important for HIV-1 transcription. Others and we previously showed that protein phosphatase-2A and protein phosphatase-1 regulates HIV-1 transcription. In the present study we analyze relative contribution of PP2A and PP1 to dephosphorylation of CDK9 and to HIV-1 transcription in vitro and in vivo.
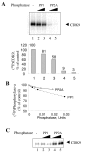
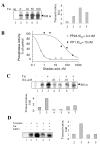
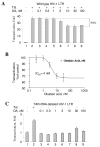
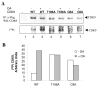
Results: In vitro, PP2A but not PP1 dephosphorylated autophosphorylated CDK9 and reduced complex formation between P-TEFb, Tat and TAR RNA. Inhibition of PP2A by okadaic acid inhibited basal as well as Tat-induced HIV-1 transcription whereas inhibition of PP1 by recombinant nuclear inhibitor of PP1 (NIPP1) inhibited only Tat-induced transcription in vitro. In cultured cells, low concentration of okadaic acid, inhibitory for PP2A, only mildly inhibited Tat-induced HIV-1 transcription. In contrast Tat-mediated HIV-1 transcription was strongly inhibited by expression of NIPP1. Okadaic acid induced phosphorylation of endogenous as well transiently expressed CDK9, but this induction was not seen in the cells expressing NIPP1. Also the okadaic acid did not induce phosphorylation of CDK9 with mutation of Thr 186 or with mutations in Ser-329, Thr-330, Thr-333, Ser-334, Ser-347, Thr-350, Ser-353, and Thr-354 residues involved in autophosphorylation of CDK9.
Conclusion: Our results indicate that although PP2A dephosphorylates autophosphorylated CDK9 in vitro, in cultured cells PP1 is likely to dephosphorylate CDK9 and contribute to the regulation of activated HIV-1 transcription.
Figures


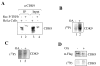
References
-
- Marcello A, Zoppe M, Giacca M. Multiple modes of transcriptional regulation by the HIV-1 Tat transactivator. IUBMB Life. 2001;51:175–181. - PubMed
-
- Liang C, Wainberg MA. The role of Tat in HIV-1 replication: an activator and/or a suppressor? AIDS Rev. 2002;4:41–49. - PubMed
-
- Kim YK, Bourgeois CF, Isel C, Churcher MJ, Karn J. Phosphorylation of the RNA polymerase II carboxyl-terminal domain by CDK9 is directly responsible for human immunodeficiency virus type 1 Tat-activated transcriptional elongation. Mol Cell Biol. 2002;22:4622–4637. doi: 10.1128/MCB.22.13.4622-4637.2002. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

