Modulation of CaV2.1 channels by the neuronal calcium-binding protein visinin-like protein-2
- PMID: 16049183
- PMCID: PMC6724833
- DOI: 10.1523/JNEUROSCI.0447-05.2005
Modulation of CaV2.1 channels by the neuronal calcium-binding protein visinin-like protein-2
Abstract
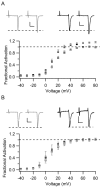
CaV2.1 channels conduct P/Q-type Ca2+ currents that are modulated by calmodulin (CaM) and the structurally related Ca2+-binding protein 1 (CaBP1). Visinin-like protein-2 (VILIP-2) is a CaM-related Ca2+-binding protein expressed in the neocortex and hippocampus. Coexpression of CaV2.1 and VILIP-2 in tsA-201 cells resulted in Ca2+ channel modulation distinct from CaM and CaBP1. CaV2.1 channels with beta2a subunits undergo Ca2+-dependent facilitation and inactivation attributable to association of endogenous Ca2+/CaM. VILIP-2 coexpression does not alter facilitation measured in paired-pulse experiments but slows the rate of inactivation to that seen without Ca2+/CaM binding and reduces inactivation of Ca2+ currents during trains of repetitive depolarizations. CaV2.1 channels with beta1b subunits have rapid voltage-dependent inactivation, and VILIP-2 has no effect on the rate of inactivation or facilitation of the Ca2+ current. In contrast, when Ba2+ replaces Ca2+ as the charge carrier, VILIP-2 slows inactivation. The effects of VILIP-2 are prevented by deletion of the CaM-binding domain (CBD) in the C terminus of CaV2.1 channels. However, both the CBD and an upstream IQ-like domain must be deleted to prevent VILIP-2 binding. Our results indicate that VILIP-2 binds to the CBD and IQ-like domains of CaV2.1 channels like CaM but slows inactivation, which enhances facilitation of CaV2.1 channels during extended trains of stimuli. Comparison of VILIP-2 effects with those of CaBP1 indicates striking differences in modulation of both facilitation and inactivation. Differential regulation of CaV2.1 channels by CaM, VILIP-2, CaBP1, and other neurospecific Ca2+-binding proteins is a potentially important determinant of Ca2+ entry in neurotransmission.
Figures







References
-
- Ames JB, Ishima R, Tanaka T, Gordon JI, Stryer L, Ikura M (1997) Molecular mechanics of calcium-myristoyl switches. Nature 389: 198-202. - PubMed
-
- Arikkath J, Campbell KP (2003) Auxiliary subunits: essential components of the voltage-gated calcium channel complex. Curr Opin Neurobiol 13: 298-307. - PubMed
-
- Bernstein HG, Baumann B, Danos P, Diekmann S, Bogerts B, Gundelfinger ED, Braunewell KH (1999) Regional and cellular distribution of neural visinin-like protein immunoreactivities (VILIP-1 and VILIP-3) in human brain. J Neurocytol 28: 655-662. - PubMed
-
- Bernstein HG, Becker A, Keilhoff G, Spilker C, Gorczyca WA, Braunewell KH, Grecksch G (2003) Brain region-specific changes in the expression of calcium sensor proteins after repeated applications of ketamine to rats. Neurosci Lett 339: 95-98. - PubMed
-
- Burgess DL, Biddlecome GH, McDonough SI, Diaz ME, Zilinski CA, Bean BP, Campbell KP, Noebels JL (1999) Beta subunit reshuffling modifies N- and P/Q-type Ca2+ channel subunit compositions in lethargic mouse brain. Mol Cell Neurosci 13: 293-311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
