Response to Staphylococcus aureus requires CD36-mediated phagocytosis triggered by the COOH-terminal cytoplasmic domain
- PMID: 16061696
- PMCID: PMC2171464
- DOI: 10.1083/jcb.200501113
Response to Staphylococcus aureus requires CD36-mediated phagocytosis triggered by the COOH-terminal cytoplasmic domain
Abstract
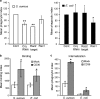
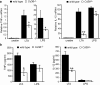
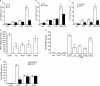
Phagocyte recognition and clearance of bacteria play essential roles in the host response to infection. In an on-going forward genetic screen, we identify the Drosophila melanogaster scavenger receptor Croquemort as a receptor for Staphylococcus aureus, implicating for the first time the CD36 family as phagocytic receptors for bacteria. In transfection assays, the mammalian Croquemort paralogue CD36 confers binding and internalization of Gram-positive and, to a lesser extent, Gram-negative bacteria. By mutational analysis, we show that internalization of S. aureus and its component lipoteichoic acid requires the COOH-terminal cytoplasmic portion of CD36, specifically Y463 and C464, which activates Toll-like receptor (TLR) 2/6 signaling. Macrophages lacking CD36 demonstrate reduced internalization of S. aureus and its component lipoteichoic acid, accompanied by a marked defect in tumor necrosis factor-alpha and IL-12 production. As a result, Cd36-/- mice fail to efficiently clear S. aureus in vivo resulting in profound bacteraemia. Thus, response to S. aureus requires CD36-mediated phagocytosis triggered by the COOH-terminal cytoplasmic domain, which initiates TLR2/6 signaling.
Figures


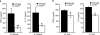
References
-
- Aitman, T.J., L.D. Cooper, P.J. Norsworthy, F.N. Wahid, J.K. Gray, B.R. Curtis, P.M. McKeigue, D. Kwiatkowski, B.M. Greenwood, R.W. Snow, et al. 2000. Malaria susceptibility and CD36 mutation. Nature. 405:1015–1016. - PubMed
-
- Akira, S., and K. Takeda. 2004. Toll-like receptor signalling. Nat. Rev. Immunol. 4:499–511. - PubMed
-
- Arbibe, L., J.P. Mira, N. Teusch, L. Kline, M. Guha, N. Mackman, P.J. Godowski, R.J. Ulevitch, and U.G. Knaus. 2000. Toll-like receptor 2-mediated NF-kappa B activation requires a Rac1-dependent pathway. Nat. Immunol. 1:533–540. - PubMed
-
- Blander, J.M., and R. Medzhitov. 2004. Regulation of phagosome maturation by signals from toll-like receptors. Science. 304:1014–1018. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

