The basal proton conductance of mitochondria depends on adenine nucleotide translocase content
- PMID: 16076285
- PMCID: PMC1316271
- DOI: 10.1042/BJ20050890
The basal proton conductance of mitochondria depends on adenine nucleotide translocase content
Abstract
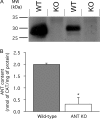
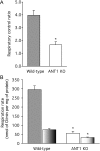
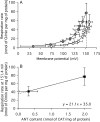
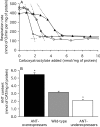
The basal proton conductance of mitochondria causes mild uncoupling and may be an important contributor to metabolic rate. The molecular nature of the proton-conductance pathway is unknown. We show that the proton conductance of muscle mitochondria from mice in which isoform 1 of the adenine nucleotide translocase has been ablated is half that of wild-type controls. Overexpression of the adenine nucleotide translocase encoded by the stress-sensitive B gene in Drosophila mitochondria increases proton conductance, and underexpression decreases it, even when the carrier is fully inhibited using carboxyatractylate. We conclude that half to two-thirds of the basal proton conductance of mitochondria is catalysed by the adenine nucleotide carrier, independently of its ATP/ADP exchange or fatty-acid-dependent proton-leak functions.
Figures

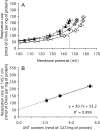


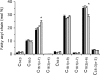

References
-
- Brand M. D., Chien L. F., Ainscow E. K., Rolfe D. F. S., Porter R. K. The causes and functions of mitochondrial proton leak. Biochim. Biophys. Acta. 1994;1187:132–139. - PubMed
-
- Brand M. D. The proton leak across the mitochondrial inner membrane. Biochim. Biophys. Acta. 1990;1018:128–133. - PubMed
-
- Rolfe D. F. S., Brand M. D. Contribution of mitochondrial proton leak to skeletal muscle respiration and to standard metabolic rate. Am. J. Physiol. 1996;271:C1380–C1389. - PubMed
-
- Rolfe D. F. S., Brand M. D. The physiological significance of mitochondrial proton leak in animal cells and tissues. Biosci. Rep. 1997;17:9–16. - PubMed
-
- Rolfe D. F. S., Newman J. M., Buckingham J. A., Clark M. G., Brand M. D. Contribution of mitochondrial proton leak to respiration rate in working skeletal muscle and liver and to SMR. Am. J. Physiol. 1999;276:C692–C699. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

