Suppression of death receptor-mediated apoptosis by 1,25-dihydroxyvitamin D3 revealed by microarray analysis
- PMID: 16093247
- PMCID: PMC3249976
- DOI: 10.1074/jbc.M506648200
Suppression of death receptor-mediated apoptosis by 1,25-dihydroxyvitamin D3 revealed by microarray analysis
Abstract
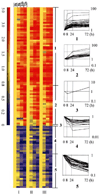
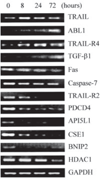
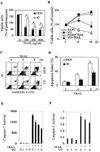
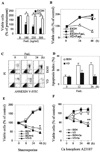
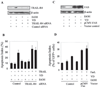
Recent studies suggest that growth inhibition by 1,25-dihydroxyvitamin D3 represents an innovative approach to ovarian cancer therapy. To understand the molecular mechanism of 1,25-dihydroxyvitamin D3 action, we profiled the hormone-induced changes in the transcriptome of ovarian cancer cells using microarray technology. More than 200 genes were identified to be regulated by 1,25-dihydroxyvitamin D3. Reverse transcription-PCR analyses confirmed the regulation of a group of apoptosis-related genes, including the up-regulation of the decoy receptor that inhibits tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) action, TRAIL receptor 4, and the down-regulation of Fas, the receptor that mediates the action of Fas ligand. The regulation was further confirmed at the protein level. Consistent with the regulation of the death receptors, pretreatment with 1,25-dihydroxyvitamin D3 decreased apoptosis induced by TRAIL and Fas ligand. Because persistent 1,25-dihydroxyvitamin D3 treatment has been shown to induce apoptosis in ovarian cancer, the hormone appears to exert a dual effect on the death of ovarian cancer cells. Knockdown of TRAIL receptor 4 by RNA interference or ectopic expression of Fas relieved the suppressive effect of 1,25-dihydroxyvitamin D3, showing that molecular manipulation of death receptors is a viable approach to overcome the protective effect of 1,25-dihydroxyvitamin D3 on the apoptosis of ovarian cancer. These strategies may allow ovarian cancer patients to benefit from therapy with both 1,25-dihydroxyvitamin D3 and ligands for death receptors, such as TRAIL, shown to selectively induce apoptosis in cancer but not normal cells.
Figures


References
-
- Webb AR, Kline L, Holick MF. J. Clin. Endocrinol. Metab. 1988;67:373–378. - PubMed
-
- Muir C, Waterhouse J, Mack T, Powell J, Whelan S. Cancer Incidence in Five Continents, International Agency for Research on Cancer Control (IARC), Scientific Publication No. 88. Vol. V. Lyon, France: IARC; 1987.
-
- Devesa SS, Grauman MA, Blot WJ, Pennello GA, Hoover RN, Fraumeni JF., Jr . Atlas of Cancer Mortality in the United States. Bethesda, MD: National Institutes of Health Publication No. 99-4564, NCI, National Institutes of Health; 1999.
-
- Jiang F, Li P, Fornace AJ, Jr, Nicosia SV, Bai W. J. Biol. Chem. 2003;278:48030–48040. - PubMed
-
- Miettinen S, Ahonen MH, Lou YR, Manninen T, Tuohimaa P, Syvala H, Ylikomi T. Int. J. Cancer. 2004;108:367–373. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

