Asymmetric cell divisions promote stratification and differentiation of mammalian skin
- PMID: 16094321
- PMCID: PMC1399371
- DOI: 10.1038/nature03922
Asymmetric cell divisions promote stratification and differentiation of mammalian skin
Abstract
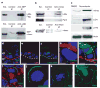
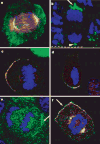
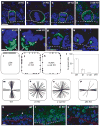
The epidermis is a stratified squamous epithelium forming the barrier that excludes harmful microbes and retains body fluids. To perform these functions, proliferative basal cells in the innermost layer periodically detach from an underlying basement membrane of extracellular matrix, move outward and eventually die. Once suprabasal, cells stop dividing and enter a differentiation programme to form the barrier. The mechanism of stratification is poorly understood. Although studies in vitro have led to the view that stratification occurs through the delamination and subsequent movement of epidermal cells, most culture conditions favour keratinocytes that lack the polarity and cuboidal morphology of basal keratinocytes in tissue. These features could be important in considering an alternative mechanism, that stratification occurs through asymmetric cell divisions in which the mitotic spindle orients perpendicularly to the basement membrane. Here we show that basal epidermal cells use their polarity to divide asymmetrically, generating a committed suprabasal cell and a proliferative basal cell. We further demonstrate that integrins and cadherins are essential for the apical localization of atypical protein kinase C, the Par3-LGN-Inscuteable complex and NuMA-dynactin to align the spindle.
Figures

Similar articles
-
Asymmetric cell divisions promote Notch-dependent epidermal differentiation.Nature. 2011 Feb 17;470(7334):353-8. doi: 10.1038/nature09793. Nature. 2011. PMID: 21331036 Free PMC article.
-
NuMA localization, stability, and function in spindle orientation involve 4.1 and Cdk1 interactions.Mol Biol Cell. 2013 Dec;24(23):3651-62. doi: 10.1091/mbc.E13-05-0277. Epub 2013 Oct 9. Mol Biol Cell. 2013. PMID: 24109598 Free PMC article.
-
AGS3 antagonizes LGN to balance oriented cell divisions and cell fate choices in mammalian epidermis.Elife. 2023 Apr 5;12:e80403. doi: 10.7554/eLife.80403. Elife. 2023. PMID: 37017303 Free PMC article.
-
Going vertical: functional role and working principles of the protein Inscuteable in asymmetric cell divisions.Cell Mol Life Sci. 2013 Nov;70(21):4039-46. doi: 10.1007/s00018-013-1319-z. Epub 2013 Mar 21. Cell Mol Life Sci. 2013. PMID: 23516018 Free PMC article. Review.
-
The basement membrane in epidermal polarity, stemness, and regeneration.Am J Physiol Cell Physiol. 2022 Dec 1;323(6):C1807-C1822. doi: 10.1152/ajpcell.00069.2022. Epub 2022 Nov 14. Am J Physiol Cell Physiol. 2022. PMID: 36374168 Review.
Cited by
-
High proliferation and delamination during skin epidermal stratification.Nat Commun. 2021 May 28;12(1):3227. doi: 10.1038/s41467-021-23386-4. Nat Commun. 2021. PMID: 34050161 Free PMC article.
-
Numb family proteins: novel players in cardiac morphogenesis and cardiac progenitor cell differentiation.Biomol Concepts. 2015 Apr;6(2):137-48. doi: 10.1515/bmc-2015-0003. Biomol Concepts. 2015. PMID: 25883210 Free PMC article. Review.
-
Cortical dynein is critical for proper spindle positioning in human cells.J Cell Biol. 2012 Oct 1;199(1):97-110. doi: 10.1083/jcb.201203166. J Cell Biol. 2012. PMID: 23027904 Free PMC article.
-
Adherens junctions and stem cells.Subcell Biochem. 2012;60:359-77. doi: 10.1007/978-94-007-4186-7_15. Subcell Biochem. 2012. PMID: 22674079 Free PMC article. Review.
-
Human tumour viruses and the deregulation of cell polarity in cancer.Nat Rev Cancer. 2012 Dec;12(12):877-86. doi: 10.1038/nrc3400. Nat Rev Cancer. 2012. PMID: 23175122 Review.
References
-
- Fuchs E, Raghavan S. Getting under the skin of epidermal morphogenesis. Nature Rev Genet. 2002;3:199–209. - PubMed
-
- Vaezi A, Bauer C, Vasioukhin V, Fuchs E. Actin cable dynamics and Rho/Rock orchestrate a polarized cytoskeletal architecture in the early steps of assembling a stratified epithelium. Dev Cell. 2002;3:367–381. - PubMed
-
- Watt FM, Green H. Stratification and terminal differentiation of cultured epidermal cells. Nature. 1982;295:434–436. - PubMed
-
- Betschinger J, Knoblich JA. Dare to be different: asymmetric cell division in Drosophila, C. elegans and vertebrates. Curr Biol. 2004;14:R674–R685. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

