Lipid composition influences the shape of human low density lipoprotein in vitreous ice
- PMID: 16094859
- PMCID: PMC1249495
- DOI: 10.1007/s11745-005-1409-x
Lipid composition influences the shape of human low density lipoprotein in vitreous ice
Abstract
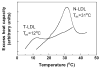
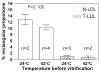
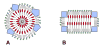
Earlier cryo-electron microscopic studies have indicated that the normal low density lipoprotein (N-LDL) has a discoid shape when its core is in the liquid-crystalline state. In the present study, we investigated whether the shape of LDL depends on the physical state and/or the lipid composition of the lipoprotein core. Using a custom-built freezing device, we vitrified NLDL samples from either above or below the phase-transition temperature of the core (42 and 24 degrees C, respectively). Cryo-electron microscopy revealed no differences between these samples and indicated a discoid shape of the N-LDL particle. In contrast, TG-enriched LDL (T-LDL) did not have discoid features and appeared to be quasi-spherical in preparations that were vitrified from either 42 or 24 degrees C. These results suggest that the shape of NLDL is discoid, regardless of the physical state of its core, whereas T-LDL is more spherical. Aspects that may influence the shape of LDL are discussed.
Figures


Similar articles
-
Preferred orientations of LDL in vitreous ice indicate a discoid shape of the lipoprotein particle.Arch Biochem Biophys. 2004 Dec 1;432(1):122-7. doi: 10.1016/j.abb.2004.08.031. Arch Biochem Biophys. 2004. PMID: 15519303
-
Structure of triglyceride-rich human low-density lipoproteins according to cryoelectron microscopy.Biochemistry. 2003 Dec 23;42(50):14988-93. doi: 10.1021/bi0354738. Biochemistry. 2003. PMID: 14674775
-
Combined data from LDL composition and size measurement are compatible with a discoid particle shape.J Lipid Res. 2004 May;45(5):954-66. doi: 10.1194/jlr.M300521-JLR200. Epub 2004 Feb 16. J Lipid Res. 2004. PMID: 14967822
-
Cryo-electron microscopy reveals human low density lipoprotein substructure.J Lipid Res. 1994 Dec;35(12):2223-31. J Lipid Res. 1994. PMID: 7897320
-
Cryoelectron microscopy of low density lipoprotein in vitreous ice.Biophys J. 1995 May;68(5):2115-23. doi: 10.1016/S0006-3495(95)80392-9. Biophys J. 1995. PMID: 7612855 Free PMC article.
Cited by
-
Core lipid, surface lipid and apolipoprotein composition analysis of lipoprotein particles as a function of particle size in one workflow integrating asymmetric flow field-flow fractionation and liquid chromatography-tandem mass spectrometry.PLoS One. 2018 Apr 10;13(4):e0194797. doi: 10.1371/journal.pone.0194797. eCollection 2018. PLoS One. 2018. PMID: 29634782 Free PMC article.
-
High hydrostatic pressure specifically affects molecular dynamics and shape of low-density lipoprotein particles.Sci Rep. 2017 Apr 6;7:46034. doi: 10.1038/srep46034. Sci Rep. 2017. PMID: 28382948 Free PMC article.
-
Enhancing the contrast of ApoB to locate the surface components in the 3D density map of human LDL.J Mol Biol. 2011 Jan 7;405(1):274-83. doi: 10.1016/j.jmb.2010.10.034. Epub 2010 Oct 26. J Mol Biol. 2011. PMID: 21029740 Free PMC article.
-
Human LDL core cholesterol ester packing: three-dimensional image reconstruction and SAXS simulation studies.J Lipid Res. 2011 Feb;52(2):256-62. doi: 10.1194/jlr.M011569. Epub 2010 Nov 3. J Lipid Res. 2011. PMID: 21047995 Free PMC article.
-
Three-dimensional cryoEM reconstruction of native LDL particles to 16Å resolution at physiological body temperature.PLoS One. 2011 May 9;6(5):e18841. doi: 10.1371/journal.pone.0018841. PLoS One. 2011. PMID: 21573056 Free PMC article.
References
-
- Hevonoja T, Pentikäinen MO, Hyvönen MT, Kovanen PT, Ala-Korpela M. Structure of Low Density Lipoprotein (LDL) Particles: Basis for Understanding Molecular Changes in Modified LDL. Biochim Biophys Acta. 2000;1488:189–210. - PubMed
-
- Segrest JP, Jones MK, De Loof H, Dashti N. Structure of Apolipoprotein B-100 in Low Density Lipoproteins. J Lipid Res. 2001;42:1346–1367. - PubMed
-
- Borén J, Ekström U, Ågren B, Nilsson-Ehle P, Innerarity TL. The Molecular Mechanism for the Genetic Disorder Familial Defective Apolipoprotein B100. J Biol Chem. 2001;276:9214–9218. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

