Modeling control strategies of respiratory pathogens
- PMID: 16102315
- PMCID: PMC3320482
- DOI: 10.3201/eid1108.040449
Modeling control strategies of respiratory pathogens
Abstract
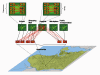
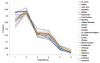
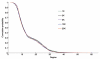
Effectively controlling infectious diseases requires quantitative comparisons of quarantine, infection control precautions, case identification and isolation, and immunization interventions. We used contact network epidemiology to predict the effect of various control policies for a mildly contagious disease, such as severe acute respiratory syndrome, and a moderately contagious disease, such as smallpox. The success of an intervention depends on the transmissibility of the disease and the contact pattern between persons within a community. The model predicts that use of face masks and general vaccination will only moderately affect the spread of mildly contagious diseases. In contrast, quarantine and ring vaccination can prevent the spread of a wide spectrum of diseases. Contact network epidemiology can provide valuable quantitative input to public health decisionmaking, even before a pathogen is well characterized.
Figures







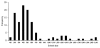
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
