Sulfiredoxin: a potential therapeutic agent?
- PMID: 16102934
- PMCID: PMC6361122
- DOI: 10.1016/j.biopha.2005.07.003
Sulfiredoxin: a potential therapeutic agent?
Abstract
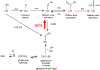
The importance of antioxidants in maintaining homeostasis has long been accepted and includes antioxidant proteins such as, peroxiredoxin (Prx), superoxide dismutase and glutathione S transferases. Sulfiredoxin (Srx) is a recently identified antioxidant protein with a role in signaling through catalytic reduction of oxidative modifications. It was first characterized for its regulation of Prx(s) through reduction of the conserved cysteine from sulfinic to sulfenic acid, thereby impacting the role of Prx in regulation of downstream transcription factors and kinase signaling pathways. Furthermore, the reduction of sulfinic to sulfenic acid prevents further oxidation of the conserved cysteine residue to sulfonic acid, the end result of which is degradation. Srx also has a role in the reduction of glutathionylation a post-translational, oxidative modification that occurs on numerous proteins and has been implicated in a wide variety of pathologies, including Parkinson's disease. The regulation of glutathionylation/deglutathionylation (or thiol switch) has been likened to phosphorylation/dephosphorylation, another post-translational modification involved in the regulation of signaling pathways. Unlike, the reduction of Prx over-oxidation, Srx-dependent deglutathionylation appears to be non-specific. Deglutathionylation of multiple proteins has been observed both in vitro and in vivo in response to oxidative and/or nitrosative stress. This review discusses Srx as a novel antioxidant, and focuses on its potential role in the regulation of glutathionylation/deglutathionylation pathways, that have been implicated in a growing number of disease states.
Figures

References
-
- Aksenov MY, Markesbery WR. Changes in thiol content and expression of glutathione redox system genes in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci Lett 2001;302(2–3): 141–5. - PubMed
-
- Barrett WC, DeGnore JP, et al. Regulation of PTP1B via glutathionylation of the active site cysteine 215. Biochemistry 1999;38(20): 6699–705. - PubMed
-
- Biteau B, Labarre J, et al. ATP-dependent reduction of cysteine–sulphinic acid by S. cerevisiae sulphiredoxin. Nature 2003; 425(6961):980–4. - PubMed
-
- Bozonet SM, Findlay VJ, et al. Oxidation of a eukaryotic 2-cys peroxiredoxin is a molecular switch controlling the transcriptional response to increasing levels of hydrogen peroxide. J Biol Chem 2005;280(24):23319–27. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

