X-ray damage to the Mn4Ca complex in single crystals of photosystem II: a case study for metalloprotein crystallography
- PMID: 16103362
- PMCID: PMC1186027
- DOI: 10.1073/pnas.0505207102
X-ray damage to the Mn4Ca complex in single crystals of photosystem II: a case study for metalloprotein crystallography
Abstract
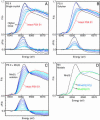
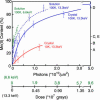
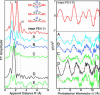
X-ray absorption spectroscopy was used to measure the damage caused by exposure to x-rays to the Mn(4)Ca active site in single crystals of photosystem II as a function of dose and energy of x-rays, temperature, and time. These studies reveal that the conditions used for structure determination by x-ray crystallography cause serious damage specifically to the metal-site structure. The x-ray absorption spectra show that the structure changes from one that is characteristic of a high-valent Mn(4)(III(2),IV(2)) oxo-bridged Mn(4)Ca cluster to that of Mn(II) in aqueous solution. This damage to the metal site occurs at a dose that is more than one order of magnitude lower than the dose that results in loss of diffractivity and is commonly considered safe for protein crystallography. These results establish quantitative x-ray dose parameters that are applicable to redox-active metalloproteins. This case study shows that a careful evaluation of the structural intactness of the active site(s) by spectroscopic techniques can validate structures derived from crystallography and that it can be a valuable complementary method before structure-function correlations of metalloproteins can be made on the basis of high-resolution x-ray crystal structures.
Figures
References
-
- Sauer, K. (1980) Acc. Chem. Res. 13, 249–256.
-
- Rutherford, A. W., Zimmermann, J.-L. & Boussac, A. (1992) in The Photosystems: Structure, Function, and Molecular Biology, ed. Barber, J. (Elsevier, Amsterdam), pp. 179–229.
-
- Debus, R. J. (1992) Biochim. Biophys. Acta 1102, 269–352. - PubMed
-
- Ort, D. R. & Yocum, C. F. (1996) Oxygenic Photosynthesis: The Light Reactions (Kluwer, Dordrecht, The Netherlands).
-
- Yachandra, V. K., Sauer, K. & Klein, M. P. (1996) Chem. Rev. 96, 2927–2950. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

