Cholera toxin subunit B inhibits IL-12 and IFN-{gamma} production and signaling in experimental colitis and Crohn's disease
- PMID: 16105891
- PMCID: PMC1774744
- DOI: 10.1136/gut.2004.062174
Cholera toxin subunit B inhibits IL-12 and IFN-{gamma} production and signaling in experimental colitis and Crohn's disease
Abstract
Background and aims: Cholera toxin B subunit (CT-B) is a powerful modulator of immune responses. The authors have previously demonstrated that oral administration of recombinant CT-B (rCT-B) is able to prevent and cure the Crohn's disease (CD)-like trinitrobenzene sulfonic acid (TNBS) mediated colitis. In this study they extended their observations and examined if rCT-B interferes with the molecular signaling underlying the Th1 type response both in TNBS colitis and in ex vivo human CD explants.
Methods: TNBS treated mice were fed with rCT-B, and IFN-gamma and IL-12 production by colonic lamina propria mononuclear cells (LPMC) was examined by ELISA. In vitro culture of mucosal explants from CD patients and non-inflammatory bowel disease controls, pre-incubated with rCT-B, were examined for IFN-gamma and IL-12 production by ELISA and semiquantitative reverse transcription polymerase chain reactions. STAT-1, -4, -6 activation and T-bet expression were examined following rCT-B treatment by western blotting both in TNBS treated mice and in human mucosal explants.
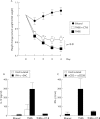
Results: rCT-B significantly reduced IL-12 and IFN-gamma secretion by LPMC from TNBS treated mice. Consistent with this, rCT-B inhibited both STAT-4 and STAT-1 activation and downregulated T-bet expression. Inhibition of Th1 signaling by CT-B associated with no change in IL-4 synthesis and expression of active STAT-6 indicating that rCT-B does not enhance Th2 cell responses. Moreover, in vitro treatment of CD mucosal explants with rCT-B resulted in reduced secretion of IL-12/IFN-gamma and inhibition of STAT-4/STAT-1 activation and T-bet expression.
Conclusions: These studies indicate that CT-B inhibits mucosal Th1 cell signaling and suggest that rCT-B may be a promising candidate for CD therapy.
Figures
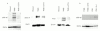


Similar articles
-
Oral administration of recombinant cholera toxin subunit B inhibits IL-12-mediated murine experimental (trinitrobenzene sulfonic acid) colitis.J Immunol. 2001 Mar 1;166(5):3522-32. doi: 10.4049/jimmunol.166.5.3522. J Immunol. 2001. PMID: 11207312
-
Regulation of the T helper cell type 1 transcription factor T-bet in coeliac disease mucosa.Gut. 2004 Aug;53(8):1090-5. doi: 10.1136/gut.2003.030551. Gut. 2004. PMID: 15247173 Free PMC article.
-
T-bet upregulation and subsequent interleukin 12 stimulation are essential for induction of Th1 mediated immunopathology in Crohn's disease.Gut. 2004 Sep;53(9):1303-8. doi: 10.1136/gut.2003.024190. Gut. 2004. PMID: 15306590 Free PMC article.
-
Reciprocal IFN-gamma and TGF-beta responses regulate the occurrence of mucosal inflammation.Immunol Today. 1997 Feb;18(2):61-4. doi: 10.1016/s0167-5699(97)01000-1. Immunol Today. 1997. PMID: 9057354 Review.
-
Role of macrophage cytokines in mucosal adjuvanticity.Adv Vet Med. 1999;41:83-104. doi: 10.1016/s0065-3519(99)80010-x. Adv Vet Med. 1999. PMID: 9890011 Review.
Cited by
-
Cholera-toxin suppresses carcinogenesis in a mouse model of inflammation-driven sporadic colon cancer.Carcinogenesis. 2015 Feb;36(2):280-90. doi: 10.1093/carcin/bgu325. Epub 2014 Dec 30. Carcinogenesis. 2015. PMID: 25550315 Free PMC article.
-
Potential roles of enteric glial cells in Crohn's disease: A critical review.Cell Prolif. 2024 Jan;57(1):e13536. doi: 10.1111/cpr.13536. Epub 2023 Aug 8. Cell Prolif. 2024. PMID: 37551711 Free PMC article. Review.
-
Therapeutic Potential of Cholera Toxin B Subunit for the Treatment of Inflammatory Diseases of the Mucosa.Toxins (Basel). 2017 Nov 23;9(12):379. doi: 10.3390/toxins9120379. Toxins (Basel). 2017. PMID: 29168738 Free PMC article. Review.
-
Phosphodiesterase 4 inhibition in the treatment of psoriasis, psoriatic arthritis and other chronic inflammatory diseases.Dermatol Ther (Heidelb). 2013 Apr 27;3(1):1-15. doi: 10.1007/s13555-013-0023-0. Print 2013 Jun. Dermatol Ther (Heidelb). 2013. PMID: 23888251 Free PMC article.
-
Human intestinal models to study interactions between intestine and microbes.Open Biol. 2020 Oct;10(10):200199. doi: 10.1098/rsob.200199. Epub 2020 Oct 21. Open Biol. 2020. PMID: 33081633 Free PMC article.
References
-
- Podolsky D. Inflammatory bowel disease. N Engl J Med 2002;347:417–29. - PubMed
-
- Fuss I, Neurath M, Boirivant M, et al. Disparate CD4+ lamina propria (LP) lymphokine secretion profiles in inflammatory bowel disease. Crohn’s disease LP cells manifest increased secretion of IFN-gamma, whereas ulcerative colitis LP cells manifest increased secretion of IL-5. J Immunol 1996;157:1261–70. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
