ATP and purinergic receptor-dependent membrane traffic in bladder umbrella cells
- PMID: 16110327
- PMCID: PMC1187935
- DOI: 10.1172/JCI24086
ATP and purinergic receptor-dependent membrane traffic in bladder umbrella cells
Abstract
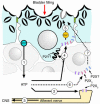
The umbrella cells that line the bladder are mechanosensitive, and bladder filling increases the apical surface area of these cells; however, the upstream signals that regulate this process are unknown. Increased pressure stimulated ATP release from the isolated uroepithelium of rabbit bladders, which was blocked by inhibitors of vesicular transport, connexin hemichannels, ABC protein family members, and nucleoside transporters. Pressure-induced increases in membrane capacitance (a measure of apical plasma membrane surface area where 1 microF approximately equals 1 cm2) were inhibited by the serosal, but not mucosal, addition of apyrase or the purinergic receptor antagonist PPADS. Upon addition of purinergic receptor agonists, increased capacitance was observed even in the absence of pressure. Moreover, knockout mice lacking expression of P2X2 and/or P2X3 receptors failed to show increases in apical surface area when exposed to hydrostatic pressure. Treatments that prevented release of Ca2+ from intracellular stores or activation of PKA blocked ATPgammaS-stimulated changes in capacitance. These results indicate that increased hydrostatic pressure stimulates release of ATP from the uroepithelium and that upon binding to P2X and possibly P2Y receptors on the umbrella cell, downstream Ca2+ and PKA second messenger cascades may act to stimulate membrane insertion at the apical pole of these cells.
Figures


Similar articles
-
Secretion of ATP from Schwann cells in response to uridine triphosphate.Eur J Neurosci. 2005 Jan;21(1):151-60. doi: 10.1111/j.1460-9568.2004.03831.x. Eur J Neurosci. 2005. PMID: 15654852
-
P2X3 knock-out mice reveal a major sensory role for urothelially released ATP.J Neurosci. 2001 Aug 1;21(15):5670-7. doi: 10.1523/JNEUROSCI.21-15-05670.2001. J Neurosci. 2001. PMID: 11466438 Free PMC article.
-
Adenosine receptor expression and function in bladder uroepithelium.Am J Physiol Cell Physiol. 2006 Aug;291(2):C254-65. doi: 10.1152/ajpcell.00025.2006. Epub 2006 Mar 29. Am J Physiol Cell Physiol. 2006. PMID: 16571869
-
Purinergic (P2) receptor control of lower genitourinary tract function and new avenues for drug action: an overview.Curr Pharm Des. 2007;13(31):3236-44. doi: 10.2174/138161207782341277. Curr Pharm Des. 2007. PMID: 18045173 Review.
-
[ATP and neuropathic pain].Seikagaku. 2004 Nov;76(11):1431-9. Seikagaku. 2004. PMID: 15626030 Review. Japanese. No abstract available.
Cited by
-
Membrane lipids and proteins as modulators of urothelial endocytic vesicles pathways.Histochem Cell Biol. 2013 Nov;140(5):507-20. doi: 10.1007/s00418-013-1095-8. Epub 2013 Apr 27. Histochem Cell Biol. 2013. PMID: 23624723 Review.
-
Modulation of bladder function by luminal adenosine turnover and A1 receptor activation.Am J Physiol Renal Physiol. 2012 Jul 15;303(2):F279-92. doi: 10.1152/ajprenal.00566.2011. Epub 2012 May 2. Am J Physiol Renal Physiol. 2012. PMID: 22552934 Free PMC article.
-
Deletion of the transient receptor potential cation channel TRPV4 impairs murine bladder voiding.J Clin Invest. 2007 Nov;117(11):3453-62. doi: 10.1172/JCI31766. J Clin Invest. 2007. PMID: 17948126 Free PMC article.
-
Pharmacology of P2X channels.Pflugers Arch. 2006 Aug;452(5):513-37. doi: 10.1007/s00424-006-0070-9. Epub 2006 Apr 29. Pflugers Arch. 2006. PMID: 16649055 Review.
-
Urothelial plaque formation in post-Golgi compartments.PLoS One. 2011;6(8):e23636. doi: 10.1371/journal.pone.0023636. Epub 2011 Aug 24. PLoS One. 2011. PMID: 21887288 Free PMC article.
References
-
- Novak I. ATP as a signaling molecule: the exocrine focus [review] News Physiol. Sci. 2003;18:12–17. - PubMed
-
- Burnstock G. Purine-mediated signalling in pain and visceral perception. Trends Pharmacol. Sci. 2001;22:182–188. - PubMed
-
- Cooke HJ, Wunderlich J, Christofi FL. “The force be with you”: ATP in gut mechanosensory transduction [review] News Physiol. Sci. 2003;18:43–49. - PubMed
-
- Sorensen CE, Novak I. Visualization of ATP release in pancreatic acini in response to cholinergic stimulus. Use of fluorescent probes and confocal microscopy. J. Biol. Chem. 2001;276:32925–32932. - PubMed
-
- Maroto R, Hamill OP. Brefeldin A block of integrin-dependent mechanosensitive ATP release from Xenopus oocytes reveals a novel mechanism of mechanotransduction. J. Biol. Chem. 2001;276:23867–23872. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

