The genomics of disulfide bonding and protein stabilization in thermophiles
- PMID: 16111437
- PMCID: PMC1188242
- DOI: 10.1371/journal.pbio.0030309
The genomics of disulfide bonding and protein stabilization in thermophiles
Abstract
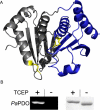
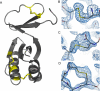
Thermophilic organisms flourish in varied high-temperature environmental niches that are deadly to other organisms. Recently, genomic evidence has implicated a critical role for disulfide bonds in the structural stabilization of intracellular proteins from certain of these organisms, contrary to the conventional view that structural disulfide bonds are exclusively extracellular. Here both computational and structural data are presented to explore the occurrence of disulfide bonds as a protein-stabilization method across many thermophilic prokaryotes. Based on computational studies, disulfide-bond richness is found to be widespread, with thermophiles containing the highest levels. Interestingly, only a distinct subset of thermophiles exhibit this property. A computational search for proteins matching this target phylogenetic profile singles out a specific protein, known as protein disulfide oxidoreductase, as a potential key player in thermophilic intracellular disulfide-bond formation. Finally, biochemical support in the form of a new crystal structure of a thermophilic protein with three disulfide bonds is presented together with a survey of known structures from the literature. Together, the results provide insight into biochemical specialization and the diversity of methods employed by organisms to stabilize their proteins in exotic environments. The findings also motivate continued efforts to sequence genomes from divergent organisms.
Figures


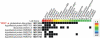

References
-
- Branden C, Tooze J. Introduction to protein structure. New York: Garland Publishing; 1991.
-
- Fahey RC, Hunt JS, Windham GC. On the cysteine and cystine content of proteins. Differences between intracellular and extracellular proteins. J Mol Evol. 1977;10:155–160. - PubMed
-
- Hiniker A, Bardwell JC. Disulfide bond isomerization in prokaryotes. Biochemistry. 2003;42:1179–1185. - PubMed
-
- Kadokura H, Katzen F, Beckwith J. Protein disulfide bond formation in prokaryotes. Annu Rev Biochem. 2003;72:111–135. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

