Proteomic identification of oxidatively modified retinal proteins in a chronic pressure-induced rat model of glaucoma
- PMID: 16123417
- PMCID: PMC11791686
- DOI: 10.1167/iovs.05-0208
Proteomic identification of oxidatively modified retinal proteins in a chronic pressure-induced rat model of glaucoma
Abstract
Purpose: Based on the evidence of an amplified production of reactive oxygen species (ROS) during glaucomatous neurodegeneration, proteomic analysis was performed to determine oxidative modification of retinal proteins after experimental elevation of intraocular pressure (IOP).
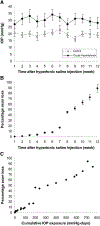
Methods: IOP elevation was induced in rats by hypertonic saline injections into episcleral veins. Protein expression was determined by two-dimensional polyacrylamide gel electrophoresis (2D-PAGE) of retinal protein lysates obtained from eyes matched for the cumulative IOP exposure and axon loss. To determine protein oxidation levels, protein carbonyls were detected through 2D-oxyblot analysis of 2,4-dinitrophenylhydrazine (DNPH)-treated samples using an anti-DNP antibody. For identification of oxidized proteins, peptide masses were analyzed by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF/MS) and liquid chromatography-tandem mass spectrometry (LC/MS/MS). In addition to use of different engines in a bioinformatic database search and performance of peptide sequencing and 2D-Western blot analysis for confirmation of the identified proteins, immunohistochemistry was used for further validation of the proteomic findings.
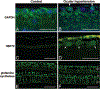
Results: Comparison of 2D-oxyblots with Coomassie Blue-stained 2D-gels revealed that approximately 60 protein spots obtained with retinal protein lysates from ocular hypertensive eyes (of >400 spots) exhibited protein carbonyl immunoreactivity, which reflects oxidatively modified proteins. There was a significant increase in anti-carbonyl reactivity in individual protein spots obtained with retinal protein lysates from ocular hypertensive eyes compared with the control (P < 0.01). The identified proteins through peptide mass fingerprinting and peptide sequencing included glyceraldehyde-3-phosphate dehydrogenase, a glycolytic enzyme; HSP72, a stress protein; and glutamine synthetase, an excitotoxicity-related protein. Immunolabeling of retina sections with specific antibodies demonstrated cellular localization of these proteins as well as retinal distribution of the increased protein carbonyl immunoreactivity in ocular hypertensive eyes.
Conclusions: The findings of this in vivo study provide novel evidence for oxidative modification of many retinal proteins in ocular hypertensive eyes and identify three specific targets of retinal protein oxidation in these eyes, thereby supporting the association of oxidative damage with neurodegeneration in glaucoma. By using a proteomic approach, this study also exemplifies that proteomics provide a very promising way to elucidate pathogenic mechanisms in glaucoma at the protein level.
Figures





References
-
- Green D, Kroemer G. The central executioners of apoptosis: caspases or mitochondria? Trends Cell Biol. 1998;8:267–271. - PubMed
-
- Cai J, Jones DP. Superoxide in apoptosis: mitochondrial generation triggered by cytochrome c loss. J Biol Chem. 1998;273:11401–11404. - PubMed
-
- Mittag TW, Danias J, Pohorenec G, et al. Retinal damage after 3 to 4 months of elevated intraocular pressure in a rat glaucoma model. Invest Ophthalmol Vis Sci. 2000;41:3451–3459. - PubMed
-
- Tatton WG, Chalmers-Redman RM, Sud A, Podos SM, Mittag TW. Maintaining mitochondrial membrane impermeability: an opportunity for new therapy in glaucoma? Surv Ophthalmol. 2001;45(suppl 3):S277–S283; discussion S295–2276. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

